













实验动物与比较医学 ›› 2026, Vol. 46 ›› Issue (1): 32-45.DOI: 10.12300/j.issn.1674-5817.2025.052
收稿日期:2025-03-27
修回日期:2025-10-29
出版日期:2026-02-25
发布日期:2026-02-14
通讯作者:
吴际(1963—),女,博士,教授,研究方向:分子生殖生物与干细胞生物。E-mail: jiwu@sjtu.edu.cn。ORCID: 0000-0002-6424-9783作者简介:徐波(1979—),男,本科,实验师,研究方向:生殖发育相关实验技术研究。E-mail:smartassi@sjtu.edu.cn。ORCID: 0009-0001-0506-0884
XU Bo1( ), CHEN Tairen2, FANG Qian1, WU Ji1(
), CHEN Tairen2, FANG Qian1, WU Ji1( )(
)( )
)
Received:2025-03-27
Revised:2025-10-29
Published:2026-02-25
Online:2026-02-14
Contact:
WU Ji (ORCID: 0000-0002-6424-9783), E-mail: jiwu@sjtu.edu.cn摘要:
目的 旨在深入探讨脊髓压迫损伤和半横断损伤小鼠模型在亚急性至慢性期(1~28 d),局部微环境中相关基因的表达次序及其分子机制,从而揭示其脊髓修复的分子特征,并为脊髓损伤治疗靶点的选择提供理论依据。 方法 选用36只8~9周龄SPF级ICR小鼠,随机分为假手术对照(CTR)组、脊髓半横断损伤(HSCI)组和脊髓压迫损伤(SCC)组,每组12只。CTR组小鼠经麻醉后,仅行椎板切除术暴露T9~T10节段的硬脊膜,并保持其完整,在空气中暴露10 min后缝合切口,不施加任何损伤干预;HSCI组小鼠在CTR组操作的基础上,通过显微器械横断70%的T9节段脊髓组织构建脊髓半横断损伤模型;SCC组小鼠也在CTR组操作的基础上,采用自制压迫器(30 g实心小铁棒)持续压迫T10节段脊髓10 min构建脊髓压迫损伤模型。在术后第1、3、7、14、21及28天,采用改良BBB(Basso-Beattie-Bresnahan)评分法评估各组小鼠运动功能的恢复情况;在术后第7、14天,将小鼠麻醉后,取损伤段脊髓组织,通过RNA测序(RNA-Seq)和富集分析来解析脊髓损伤小鼠模型中特异的分子网络演变,并利用实时荧光定量PCR法验证关键基因的表达情况。 结果 BBB评分结果显示,SCC组小鼠的运动功能恢复情况显著优于HSCI组,其BBB评分在4周内呈持续上升趋势且高于HSCI组(P<0.001)。基于RNA-Seq差异表达基因的基因本体(gene ontology,GO)和京都基因与基因组数据库(Kyoto Encyclopedia of Genes and Genomes,KEGG)富集分析显示:与CTR组相比,SCC组在术后第7天,细胞外基质相关基因显著上调(P<0.05),而轴突引导相关基因显著下调(P<0.05);在术后第21天,SCC组免疫调控和视黄醇信号通路相关基因被显著激活(P<0.05)。相比之下,HSCI组在术后第7天,炎症和免疫反应基因显著上调(P<0.001),神经元分化和突触形成相关基因则显著下调(P<0.001);在术后第21天,细胞-基质连接和N-甲基-D-天门冬氨酸受体相关基因显著上调(P<0.001)。此外,与SCC组相比,HSCI组在损伤后第7天和第21天的GO和KEGG富集分析中表现出不同的通路富集特征。术后第7天,HSCI组中的NOD样受体信号通路以及补体和凝血级联反应相关基因显著上调(P<0.001);而在术后第21天,细胞外基质-受体相互作用和神经活性配体-受体相互作用通路相关基因被显著激活(P<0.001)。最后,实时荧光定量PCR验证结果与RNA-Seq结果高度一致,进一步确认了SCC组和HSCI组关键基因表达趋势的差异。 结论 脊髓压迫损伤与脊髓半横断损伤模型可能驱动了不同的修复路径:SCC模型中部分轴突的保留使其更倾向于组织修复,而脊髓半横断损伤模型则需协调更复杂的分子网络以达成新的平衡。这一发现进一步深化了对脊髓损伤异质性调控机制的理解。
中图分类号:
徐波,陈泰任,方乾,等. 脊髓压迫和半横断损伤小鼠模型的时空基因表达谱及修复机制研究[J]. 实验动物与比较医学, 2026, 46(1): 32-45. DOI: 10.12300/j.issn.1674-5817.2025.052.
XU Bo,CHEN Tairen,FANG Qian,et al. Research on Spatiotemporal Gene Expression Profiles and Repair Mechanisms of Spinal Cord Compression and Hemisection Spinal Cord Injury Mouse Models[J]. Laboratory Animal and Comparative Medicine, 2026, 46(1): 32-45. DOI: 10.12300/j.issn.1674-5817.2025.052.
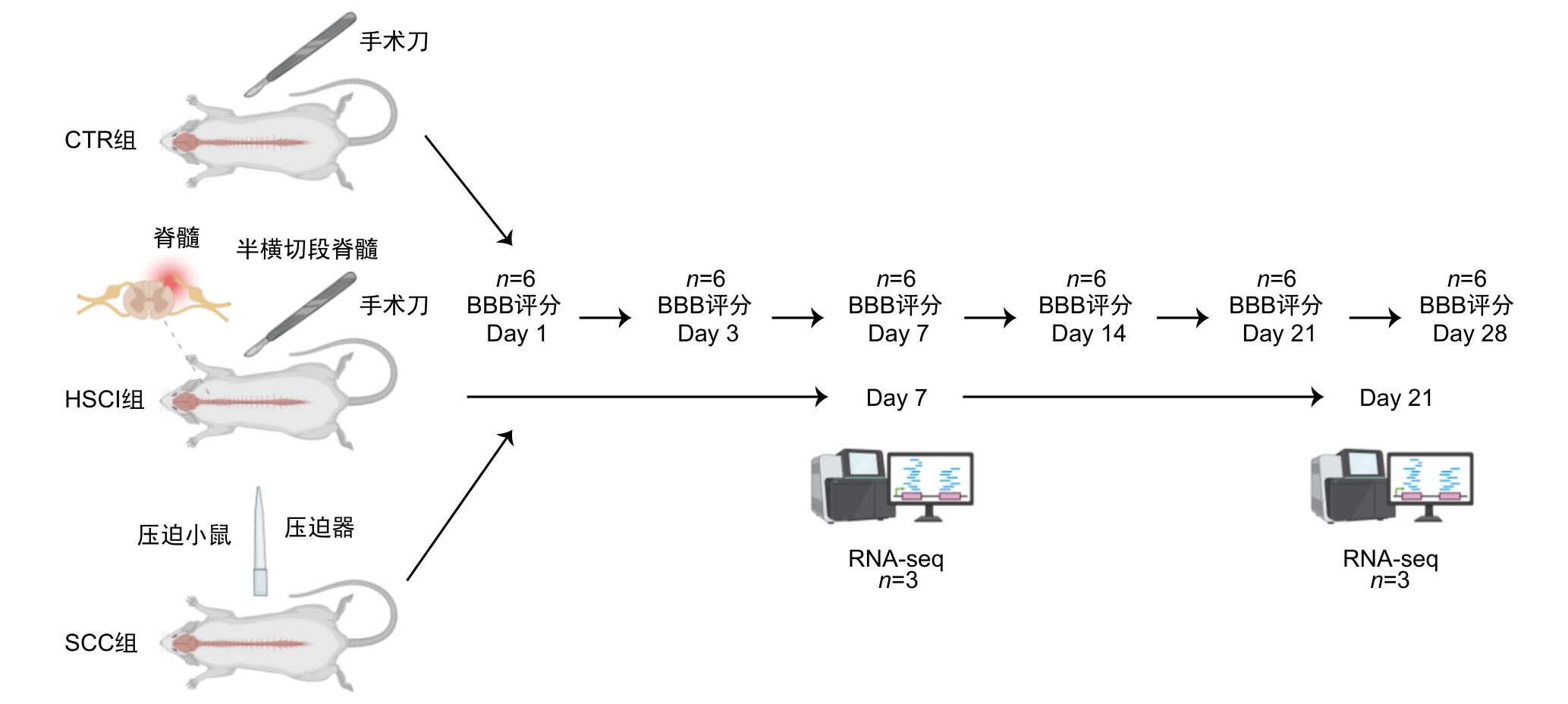
图1 模型构建和实验过程的技术路线图注:在术后第1、3、7、21和28天采用改良BBB(Basso-Beattie-Bresnahan)评分评估假手术对照(CTR)组,脊髓半横断损伤(HSCI)组和脊髓压迫损伤(SCC)组小鼠运动功能的恢复情况,并在术后第7和21天对三组小鼠脊髓损伤段组织进行RNA测序(RNA-seq)。
Figure 1 Technical roadmap for model construction and experimental processNote: On postoperative days 1, 3, 7, 21, and 28, the modified Basso-Beattie-Bresnahan scale was used to assess motor recovery in the sham-operated control (CTR) group, hemisection spinal cord injury (HSCI) group, and spinal cord compression injury (SCC) group. RNA-seq was performed on spinal cord tissue from the injured segments of mice in all three groups at days 7 and 21 post-surgery.
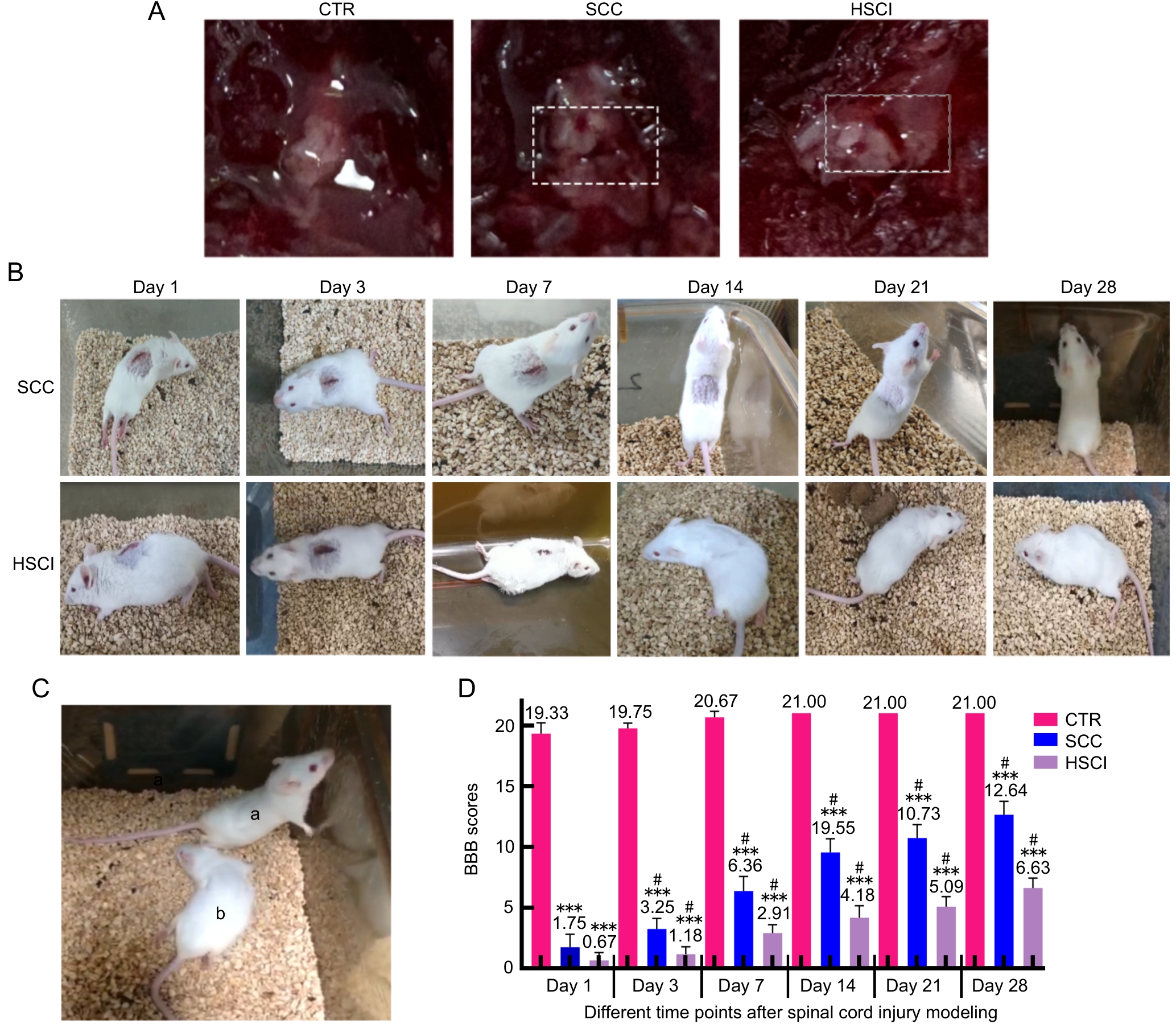
图2 假手术对照组、脊髓半横断损伤组和脊髓压迫损伤组小鼠宏观形态、恢复过程及运动功能的比较注:A,CTR组、SCC组和HSCI组小鼠的脊髓体视镜明场图(白色虚线框内为脊髓造模状态);B,SCC组和HSCI组小鼠在术后第1、3、7、14、21和28天恢复情况;C,为术后第28天SCC组(标记为a)和HSCI组(标记为b)小鼠恢复情况的对比图;D,脊髓损伤术后不同时间点的BBB评分。CTR,假手术对照组;SCC,脊髓压迫损伤组;HSCI;脊髓半横断损伤组。???与CTR组相比,P<0.001;#与组内前一个时间点相比,P<0.05。
Figure 2 Comparison of macroscopic morphology, recovery process and motor function of mice in sham-operated control group, spinal cord compression and hemisection spinal cord injury groupNote: A, the spinal cord stereoscopy bright-field images of CTR group, SCC group and HSCI group, respectively(the white dashed box indicates the site of spinal cord modeling); B, recovery of mice in SCC and HSCI groups at 1, 3, 7, 14, 21 and 28 days after modeling; C, the comparison chart of the recovery of mice in the SCC group (marked as a) and the HSCI group (marked as b) on the 28th day after surgery; D, BBB scores at different time points after SCI modeling. CTR, sham-operated control group; SCC, spinal cord compression injury group; HSCI, hemisection spinal cord injury group. ???P<0.001, compared with CTR group; #P<0.05, compared with the previous time point within the group.
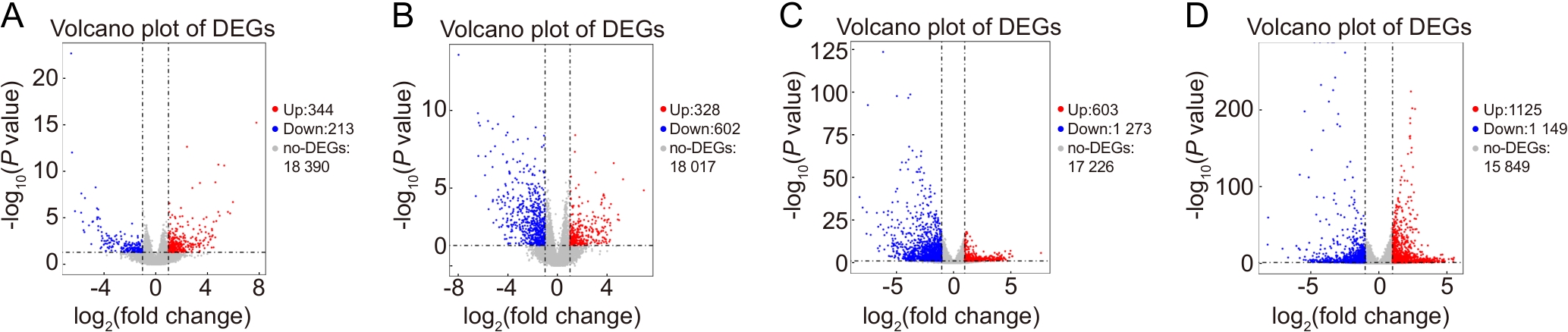
图3 脊髓损伤组小鼠与假手术对照组小鼠在第7天和第21天基因表达差异的火山图注:A~B,与CTR组相比,SCC组小鼠在术后第7天和第21天基因的表达情况;C~D,与CTR组相比,HSCI组小鼠在第7天和第21天基因的表达情况。红色圆点代表上调的差异表达基因(DEGs),蓝色圆点代表下调的DEGs,灰色点代表表达无显著差异的基因。CTR,假手术对照组;HSCI,脊髓半横断损伤组;SCC,脊髓压迫损伤组。
Figure 3 Volcano plots of differentially expressed genes in spinal cord injury models compared with the sham-operated control group at day 7 and day 21 post-injuryNote: A-B, gene expression in the SCC group compared with the CTR group at postoperative day 7 and day 21; C-D, gene expression in the HSCI group compared with the CTR group at day 7 and day 21. Red dots represent up-regulated differentially expressed genes (DEGs), blue dots represent down-regulated DEGs, and gray dots represent genes with no significant difference in expression. CTR, sham-operated control group; HSCI, hemisection spinal cord injury group; SCC, spinal cord compression injury group.
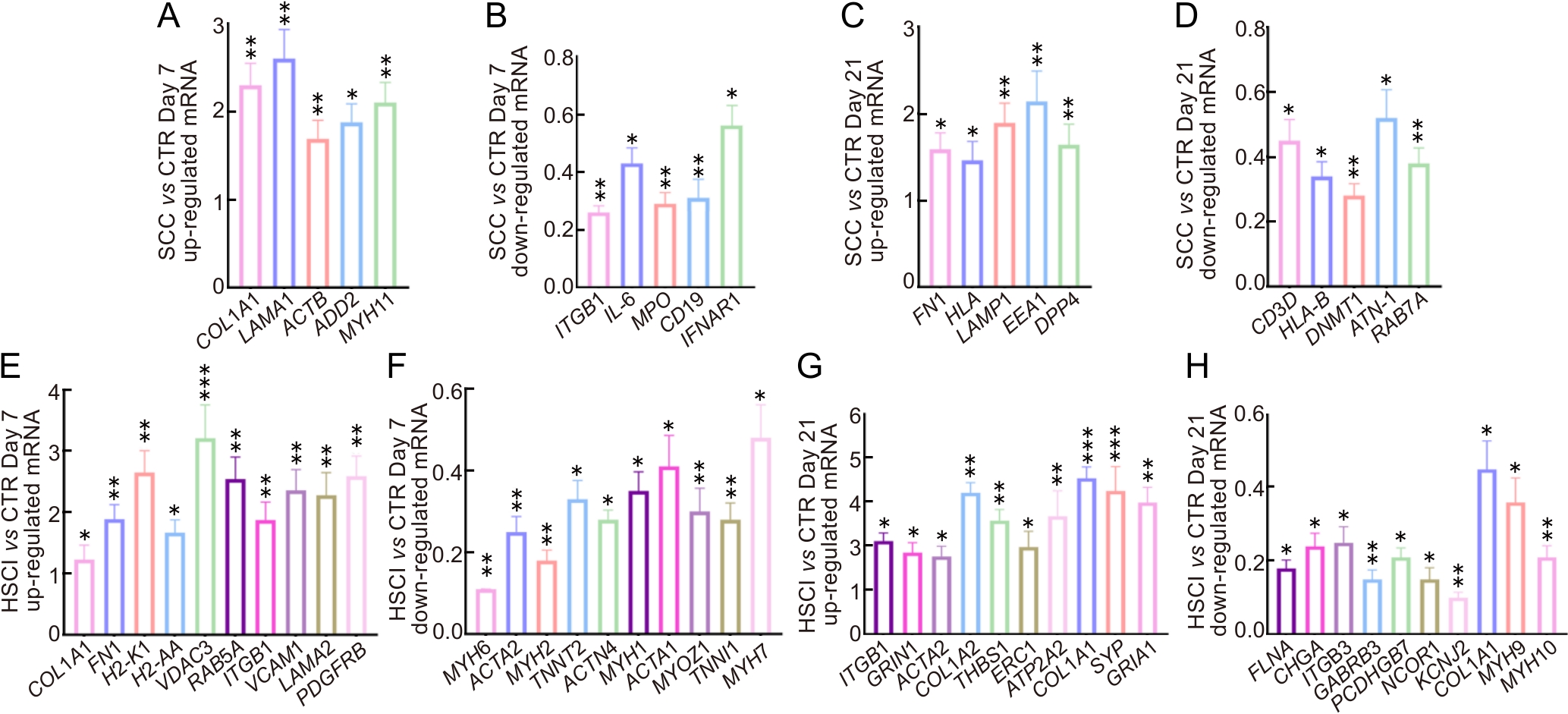
图4 实时荧光定量PCR验证表达差异基因的mRNA相对表达水平注:A~D,与CTR组相比,SCC组在第7和21天上调和下调关键基因的实时荧光定量PCR结果;E~H,与CTR组相比,HSCI组在第7和21天上调和下调关键基因的实时荧光定量PCR结果。CTR,假手术对照组;SCC,脊髓压迫损伤组;HSCI,脊髓半横断损伤组。图中数据均以CTR组基因表达水平为1进行归一化展示。实时荧光定量PCR实验使用RNA测序后剩余的同一批小鼠组织样本进行检测(n=3)。?P<0.05,??P<0.01,???P<0.001。
Figure 4 Validation of mRNA expression levels of differentially expressed genes by real time fluorogenic quantitative PCRNote: A-D, real time fluorogenic quantitative PCR results of the key genes that up-regulated and down-regulated in the SCC group compared with the CTR group at days 7 and 21; E-H, real time fluorogenic quantitative PCR results of the key genes that up-regulated and down-regulated at days 7 and 21 in the HSCI group compared with the CTR group. CTR, sham-operated control group; SCC, spinal cord contusion injury group; HSCI, hemisection spinal cord injury group. All data in the figure are normalized to a baseline of 1 for the CTR group. Real time fluorogenic quantitative PCR experiments were performed using the same batch of mouse tissue samples remaining after RNA-seq(n=3). ?P<0.05, ??P<0.01, ???P<0.001.

图5 脊髓压迫损伤组与假手术对照组小鼠在术后第7天和第21天差异表达基因的富集分析结果注:A~B,术后第7天,SCC组和CTR组之间上调和下调差异表达基因的基因本体(GO)富集分析结果;C~D,术后第7天,SCC组和CTR组之间上调和下调差异表达基因的京都基因与基因组数据库(KEGG)富集分析结果;E~F,术后第21天,SCC组和CTR组之间上调和下调差异表达基因的GO富集分析结果;G~H,术后第21天,SCC组和CTR组之间上调和下调差异表达基因的KEGG富集分析结果。图中条形颜色代表对应功能类别的P值,反映富集显著性。SCC,脊髓压迫损伤组;CTR,假手术对照组。
Figure 5 Enrichment analysis of differentially expressed genes in the spinal cord compression group compared to the sham-operated control group at postoperative days 7 and 21Note: A-B, gene ontology (GO) enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the SCC and CTR groups on postoperative day 7; C-D, Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the SCC and CTR groups on postoperative day 7; E-F, GO enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the SCC and CTR groups on postoperative day 21; G-H, KEGG enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the SCC and CTR groups on postoperative day 21. The color of the bars represents the P value of the corresponding functional category, reflecting the significance of enrichment. SCC, spinal cord compression group; CTR, sham-operated control group.
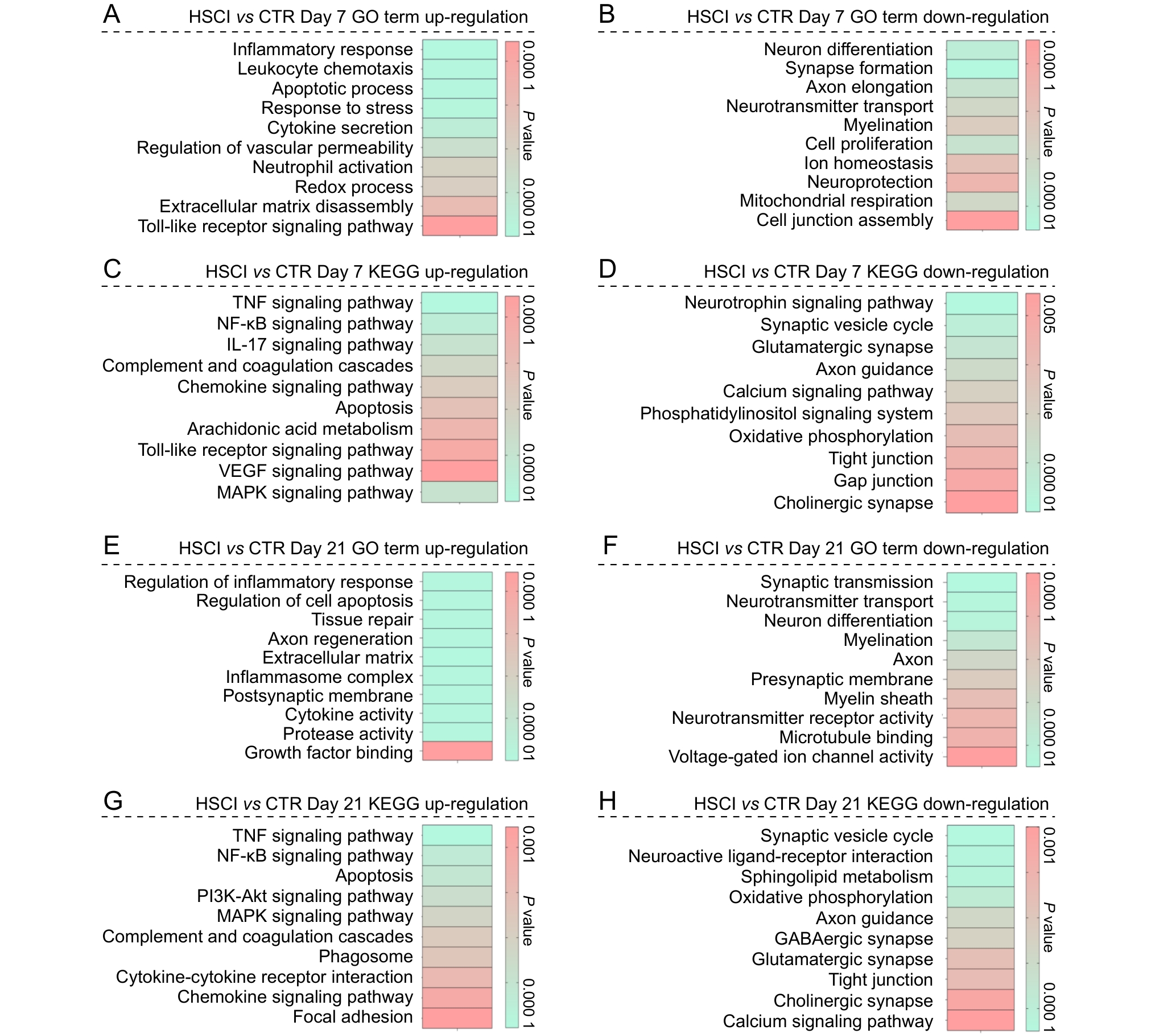
图6 脊髓半横断损伤组与假手术对照组小鼠在第7天与第21天的差异表达基因的功能富集分析结果注:A~B,术后第7天,HSCI组和CTR组之间上调和下调差异表达基因的基因本体(GO)富集分析结果;C~D,术后第7天,HSCI组和CTR组之间上调和下调差异表达基因的京都基因与基因组数据库(KEGG)富集分析结果;E~F,术后第21天,HSCI组和CTR组之间上调和下调差异表达基因的GO富集分析结果;G~H,术后第21天,HSCI组和CTR组之间上调和下调差异表达基因的KEGG富集分析结果。图中条形颜色代表对应功能类别的P值,反映富集显著性。HSCI组,脊髓半横断损伤组;CTR组,假手术对照组。
Figure 6 Functional enrichment analysis of differentially expressed genes between the hemisection spinal cord injury group and the sham-operated control group at postoperative days 7 and 21Note: A-B, gene ontology (GO) enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the HSCI and CTR groups on postoperative day 7; C-D, Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the HSCI and CTR groups on postoperative day 7; E-F, GO enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the HSCI and CTR groups on postoperative day 21; G-H, KEGG enrichment analysis results of up-regulated and down-regulated differentially expressed genes between the HSCI and CTR groups on postoperative day 21. The color of the bars represents the P value of the corresponding functional category, reflecting the significance of enrichment. HSCI, hemisection spinal cord injury group; CTR, sham-operated control group.
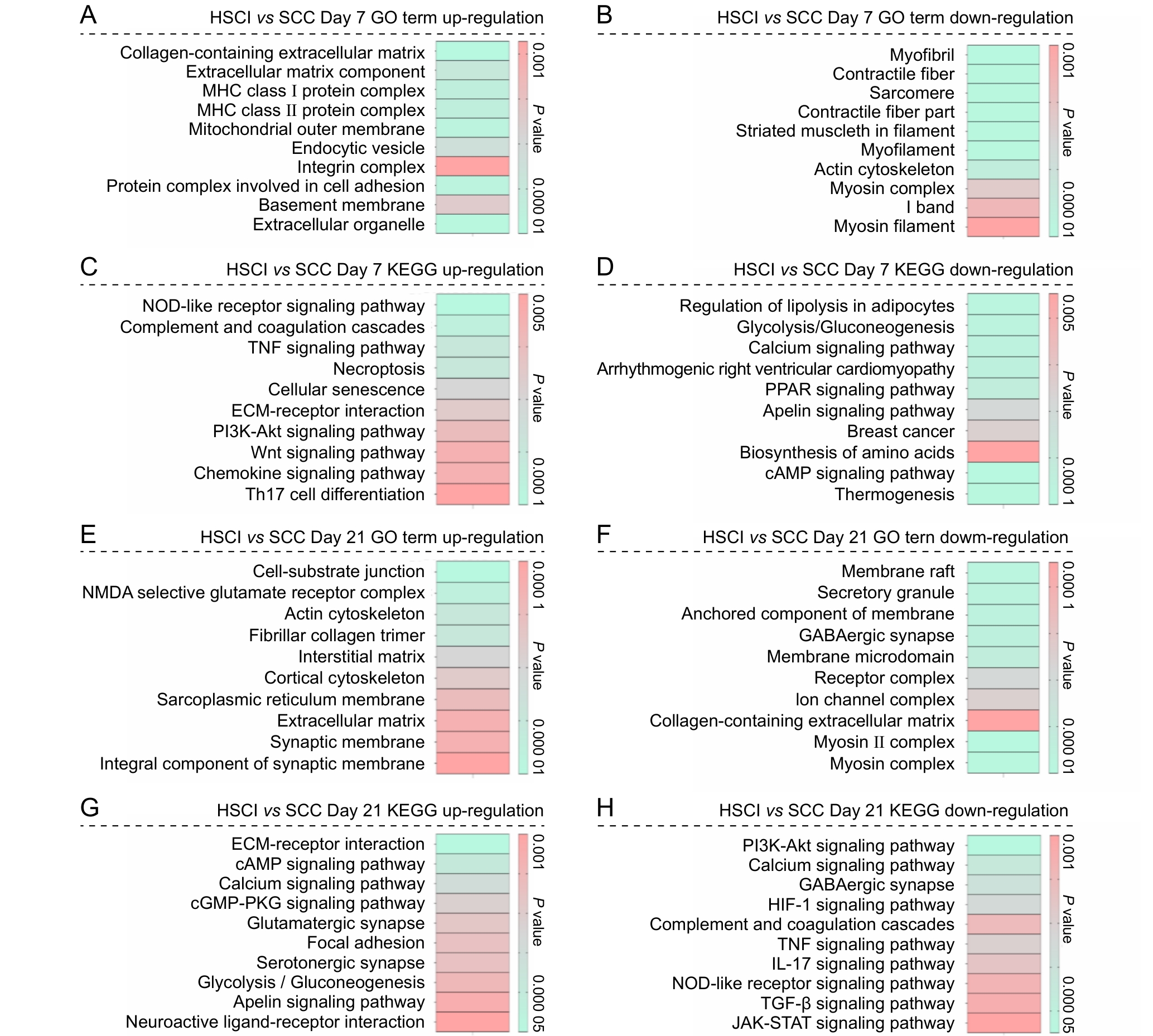
图7 脊髓半横断损伤组与脊髓压迫损伤组在术后第7天与第21天表达差异基因的功能富集分析结果注:A~B,术后第7天,HSCI组和SCC组之间上调和下调差异表达基因的基因本体(GO)富集分析结果;C~D,术后第7天,HSCI组和SCC组之间上调和下调差异表达基因的京都基因与基因组数据库(KEGG)富集分析结果;E~F,术后第21天,HSCI组和SCC组之间上调和下调差异表达基因的GO富集分析结果;G~H,术后第21天,HSCI组和SCC组之间上调和下调差异表达基因的KEGG通路富集分析结果。条形颜色表示相应功能条目的P值,用于反映富集的显著性水平。HSCI组,脊髓半横断损伤组;SCC组,脊髓压迫损伤组。
Figure 7 Functional enrichment analysis of differentially expressed genes between the hemisection spinal cord injury group and the spinal cord compression injury group at postoperative days 7 and 21Note: A-B, gene ontology (GO) enrichment analysis results of up?regulated and down?regulated differentially expressed genes between the HSCI and SCC groups on postoperative day 7; C-D, Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analysis results of up?regulated and down?regulated differentially expressed genes between the HSCI and SCC groups on postoperative day 7; E-F, GO enrichment analysis results of up?regulated and down?regulated differentially expressed genes between the HSCI and SCC groups on postoperative day 21; G-H, KEGG pathway enrichment analysis results of up?regulated and down?regulated differentially expressed genes between the HSCI and SCC groups on postoperative day 21. The color of the bars represents the P value of the corresponding functional term, indicating the significance level of enrichment. HSCI, hemisection spinal cord injury group; SCC, spinal cord compression group.
| [1] | 崔英杰, 孙文卓, 张红梅, 等. 颈椎椎间盘突出致脊髓半横断综合征误诊为脑卒中1例报告[J]. 脊柱外科杂志, 2022, 20(4): 283-285. DOI: 10.3969/j.issn.1672-2957.2022.04.013 . |
| CUI Y J, SUN W Z, ZHANG H M, et al. Brown-Sequard syndrome due to cervical disc herniation misdiagnosed as stroke: a case report[J]. J Spinal Surg, 2022, 20(4): 283-285. DOI: 10.3969/j.issn.1672-2957.2022.04.013 . | |
| [2] | COWAN H, LAKRA C, DESAI M. Autonomic dysreflexia in spinal cord injury[J]. Bmj, 2020: m3596. DOI:10.1136/bmj.m3596 . |
| [3] | FAN B Y, WEI Z J, YAO X, et al. Microenvironment imbalance of spinal cord injury[J]. Cell Transplant, 2018, 27(6):853-866. DOI:10.1177/0963689718755778 . |
| [4] | BASSO D M, FISHER L C, ANDERSON A J, et al. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains[J]. J Neurotrauma, 2006, 23(5):635-659. DOI:10.1089/neu.2006. 23.635 . |
| [5] | ASHAMMAKHI N, KIM H J, EHSANIPOUR A, et al. Regenerative therapies for spinal cord injury[J]. Tissue Eng Part B Rev, 2019, 25(6):471-491. DOI:10.1089/ten.teb.2019.0182 . |
| [6] | DAVID G, PFYFFER D, VALLOTTON K, et al. Longitudinal changes of spinal cord grey and white matter following spinal cord injury[J]. J Neurol Neurosurg Psychiatry, 2021, 92(11):1222-1230. DOI:10.1136/jnnp-2021-326337 . |
| [7] | ZÜCHNER M, GLOVER J C, BOULLAND J L. A neonatal mouse spinal cord compression injury model[J]. JoVE, 2016(109): 1-10. DOI:10.3791/53498 |
| [8] | ZHU S P, YING Y B, WU Q J, et al. Alginate self-adhesive hydrogel combined with dental pulp stem cells and FGF21 repairs hemisection spinal cord injury via apoptosis and autophagy mechanisms[J]. Chem Eng J, 2021, 426:130827. DOI:10.1016/j.cej.2021.130827 . |
| [9] | EVANS R M, DAVIES M. ScoreCentre: a computer program to assist with collection and calculation of BBB locomotor scale data[J]. J Neurosci Methods, 2010, 194(1):102-107. DOI:10.1016/j.jneumeth.2010.09.018 . |
| [10] | OTANI M, KUSHIDA Y, KURODA Y, et al. New rat model of spinal cord infarction with long-lasting functional disabilities generated by intraspinal injection of endothelin-1[J]. Stroke Vasc Neurol, 2025, 10(2): 1-10. DOI:10.1136/svn-2023-002962 . |
| [11] | WILKINS N, SKINNER N P, MOTOVYLYAK A, et al. Evolution of magnetic resonance imaging as predictors and correlates of functional outcome after spinal cord contusion injury in the rat[J]. J Neurotrauma, 2020, 37(6):889-898. DOI:10.1089/neu.2019.6731 . |
| [12] | NING X J, LU X H, LUO J C, et al. Molecular mechanism of microRNA-21 promoting Schwann cell proliferation and axon regeneration during injured nerve repair[J]. RNA Biol, 2020, 17(10):1508-1519. DOI:10.1080/15476286.2020.1777767 . |
| [13] | MAGGIORE J C, BURRELL J C, BROWNE K D, et al. Tissue engineered axon-based "living scaffolds" promote survival of spinal cord motor neurons following peripheral nerve repair[J]. J Tissue Eng Regen Med, 2020, 14(12):1892-1907. DOI:10.1002/term.3145 . |
| [14] | HU X, XU W, REN Y L, et al. Spinal cord injury: molecular mechanisms and therapeutic interventions[J]. Signal Transduct Target Ther, 2023, 8:1-28. DOI:10.1038/s41392-023-01477-6 . |
| [15] | FAN L, LIU C, CHEN X X, et al. Exosomes-loaded electroconductive hydrogel synergistically promotes tissue repair after spinal cord injury via immunoregulation and enhancement of myelinated axon growth[J]. Adv Sci, 2022, 9(13):1-22. DOI:10.1002/advs.202105586 . |
| [16] | QIAN D F, XU J Q, ZHANG X L, et al. Microenvironment self-adaptive nanomedicine promotes spinal cord repair by suppressing inflammation cascade and neural apoptosis[J]. Adv Mater, 2024, 36(50): e2307624. DOI:10.1002/adma. 202307624 . |
| [17] | MA J J, LI J Y, WANG X R, et al. GDNF-loaded polydopamine nanoparticles-based anisotropic scaffolds promote spinal cord repair by modulating inhibitory microenvironment[J]. Adv Healthc Mater, 2023, 12(8): e2202377. DOI:10.1002/adhm.202202377 . |
| [18] | GAO L S, WANG C, QIN B, et al. 6-phosphofructo-2-kinase/fructose-2, 6-bisphosphatase Suppresses Neuronal Apoptosis by Increasing Glycolysis and "cyclin-dependent kinase 1-Mediated Phosphorylation of p27 After Traumatic Spinal Cord Injury in Rats[J]. Cell Transplant, 2020, 29:1-14. DOI:10.1177/0963689720950226 . |
| [19] | MATA-BERMUDEZ A, RÍOS C, BURELO M, et al. Amantadine prevented hypersensitivity and decreased oxidative stress by NMDA receptor antagonism after spinal cord injury in rats[J]. Eur J Pain, 2021, 25(8):1839-1851. DOI:10.1002/ejp.1795 . |
| [20] | ZHOU W J, ZHAO T T, DU J Y, et al. TIGAR promotes neural stem cell differentiation through acetyl-CoA-mediated histone acetylation[J]. Cell Death Dis, 2019, 10:1-13. DOI:10.1038/s41419-019-1434-3 . |
| [21] | ZHAO J, CHEN K Z, WANG T, et al. Vimentin inhibits neuronal apoptosis after spinal cord injury by enhancing autophagy[J]. CNS Neurosci Ther, 2025, 31(1): 1-14. DOI:10.1111/cns.70200 . |
| [22] | YAO R B, REN L R, WANG S Y, et al. Euxanthone inhibits traumatic spinal cord injury via anti-oxidative stress and suppression of p38 and PI3K/Akt signaling pathway in a rat model[J]. Transl Neurosci, 2021, 12(1):114-126. DOI:10.1515/tnsci-2021-0012 . |
| [23] | CAO J F, HU X, XIONG L, et al. Interference of interleukin-1β mediated by lentivirus promotes functional recovery of spinal cord contusion injury in rats via the PI3K/AKT1 signaling pathway[J]. Mediat Inflamm, 2022, 2022(1):1-17. DOI:10.1155/2022/6285099 . |
| [24] | WANG Y M, ZHAO Z, REGE S V, et al. 3K3A-activated protein C stimulates postischemic neuronal repair by human neural stem cells in mice[J]. Nat Med, 2016, 22(9):1050-1055. DOI:10.1038/nm.4154 . |
| [1] | 何家祺, 周园园, 聂永强, 王朝霞, 徐汪节. 香烟烟雾暴露诱导慢性阻塞性肺疾病小鼠模型的构建及多维度病理评估[J]. 实验动物与比较医学, 2026, 46(1): 11-19. |
| [2] | 贡磊磊, 王晓霞, 封学伟, 李心蕾, 赵涵, 张雪艳, 冯欣. 不同浓度环磷酰胺诱导早发性卵巢功能不全小鼠模型及作用机制研究[J]. 实验动物与比较医学, 2025, 45(4): 403-410. |
| [3] | 姜娟, 宋宁, 连文博, 邵丛丛, 顾文文, 石燕. 两种浓度乙醇溶液灌注建立小鼠宫腔粘连模型的组织病理和分子病理表型比较[J]. 实验动物与比较医学, 2025, 45(4): 393-402. |
| [4] | 罗莲莲, 袁艳春, 王俊岭, 时广森. 肌萎缩侧索硬化症小鼠模型研究进展[J]. 实验动物与比较医学, 2025, 45(3): 290-299. |
| [5] | 潘钱家, 葛峻沂, 胡楠, 华飞, 顾敏. 基于16S rRNA测序的2型糖尿病db/db小鼠模型口腔菌群差异分析[J]. 实验动物与比较医学, 2025, 45(2): 147-157. |
| [6] | 戚龙菊, 陈世园, 廖泽华, 石袁虎, 孙郁雨, 王庆华. 经血干细胞移植联合运动训练促进大鼠脊髓损伤康复的转录组学分析[J]. 实验动物与比较医学, 2024, 44(5): 531-542. |
| [7] | 马婧威, 李根, 杨杨, 臧彩霞, 鲍秀琦, 张丹. 环磷酰胺诱导生精障碍小鼠模型不同恢复期的比较研究[J]. 实验动物与比较医学, 2023, 43(2): 112-123. |
| [8] | 任晨吟, 高思琦, 李浩, 唐标, 杨华, 刘月环. 两株CRISPR工程大肠埃希菌在小鼠体内的定植实验研究[J]. 实验动物与比较医学, 2022, 42(6): 541-550. |
| [9] | 于士颜. 小鼠模型在消化道黏膜免疫及感染性疾病研究中的应用进展[J]. 实验动物与比较医学, 2022, 42(1): 3-10. |
| [10] | 颜义彤, 白帆, 荆瀛黎, 王立淼, 李子晗, 于艳. 小鼠脊髓损伤打击模型精细行为的比较研究[J]. 实验动物与比较医学, 2021, 41(4): 290-298. |
| [11] | 姚玎, 周晶, 严国锋, 王会阳, 汪雅荻, 马政文. 盐敏感性高血压小鼠模型的建立[J]. 实验动物与比较医学, 2020, 40(4): 314-. |
| [12] | 柴文君, 孙 磊, 刘晓丽, 潘洪玉, 郭天安, 徐 烨, 闫明霞. 超声引导下左心室内注射人肺癌细胞建立骨转移小鼠模型[J]. 实验动物与比较医学, 2020, 40(3): 183-. |
| [13] | 蒋红利, 马红叶, 薛瑾虹, 孙凌霜, 陈蕾. 绒毛蛋白1在单侧肾切除Habu肾炎小鼠模型中的作用[J]. 实验动物与比较医学, 2020, 40(1): 1-8. |
| [14] | 李慧. Foxp2小鼠模型中发育性言语障碍的分子遗传学研究[J]. 实验动物与比较医学, 2019, 39(4): 331-336. |
| [15] | 何一旻, 顾鸣敏. Myh13敲除小鼠表型的初步分析[J]. 实验动物与比较医学, 2019, 39(3): 193-200. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||