













实验动物与比较医学 ›› 2026, Vol. 46 ›› Issue (1): 20-31.DOI: 10.12300/j.issn.1674-5817.2025.034
收稿日期:2025-03-04
修回日期:2025-06-13
出版日期:2026-02-25
发布日期:2026-02-14
通讯作者:
白鹰(1965—),女,博士,教授,主任医师,研究方向:缺血性脑卒中与神经保护。E-mail: 1029981825@qq.com。ORCID:0009-0008-1965-8801作者简介:徐英韬(1997—),男,硕士,住院医师,研究方向:缺血性脑卒中与神经保护。E-mail: 951072549@qq.com
基金资助:
XU Yingtao, WANG Mengmeng, LIN Ping, CHI Haitao, WANG Yi, BAI Ying( )
)
Received:2025-03-04
Revised:2025-06-13
Published:2026-02-25
Online:2026-02-14
Contact:
BAI Ying (ORCID: 0009-0008-1965-8801), E-mail: 1029981825@qq.com摘要:
目的 通过电凝法阻断小鼠大脑中动脉构建中动脉闭塞(middle cerebral artery occlusion,MCAO)模型,研究人羊膜间充质干细胞(human amniotic mesenchymal stem cells,hAMSCs)来源的外泌体(exosome,EXO)改善缺血性脑卒中及调控神经细胞铁死亡(ferroptosis)损伤的作用机制。 方法 将32只6~8周龄SPF级雄性C57BL/6J小鼠随机分为假手术组(Sham)、模型组(MCAO)、模型+生理盐水组(MCAO+NaCl)、模型+外泌体组(MCAO+EXO),每组8只。使用电凝法建立小鼠大脑MCAO模型,Sham组暴露大脑中动脉但不实施电凝;在电凝造模24 h前,向MCAO+EXO组小鼠尾静脉注射100 μL来自hAMSCs培养上清液的外泌体(9.5×1011个/mL),向MCAO+NaCl组小鼠尾静脉注射同体积生理盐水。造模24 h后,采用Longa神经功能缺损评分法评价各组小鼠的运动神经功能损害程度;通过2,3,5-三苯基氯化四氮唑染色法评估各组小鼠脑梗死体积占比差异;通过HE染色法评估各组小鼠缺血部位脑组织中神经细胞形态学差异。通过微量比色法评估各组小鼠脑梗死区及其周围组织中亚铁离子(Fe2+)、丙二醛(malondialdehyde,MDA)、总谷胱甘肽(total glutathione,total GSH)、氧化型谷胱甘肽(oxidized glutathione,GSSG)和还原型谷胱甘肽(glutathione,GSH)的含量差异。通过实时荧光定量PCR法检测各组小鼠脑梗死区及其周围组织中铁死亡相关因子包括核因子-红细胞系2相关因子2(nuclear factor-erythroid 2-related factor 2,NRF2)、溶质载体家族7成员11(solute carrier family 7 member 11,SLC7A11)、谷胱甘肽过氧化物酶4(glutathione peroxidase 4,GPX4)的mRNA表达水平。通过蛋白质印迹法检测各组小鼠脑梗死区及其周围组织中NRF2、SLC7A11和GPX4蛋白表达水平。 结果 相较于MCAO组,MCAO+EXO组的Longa评分显著降低(P<0.01)。MCAO组小鼠脑组织出现明显梗死灶,MCAO+EXO组相较MCAO组的脑梗死体积占比显著减少(P<0.001)。与Sham组相比,MCAO组出现神经细胞空泡变性,细胞核固缩、碎裂,细胞核结构不清晰,神经细胞排列杂乱。相较于MCAO组,MCAO+EXO组小鼠的神经细胞结构较为完整,细胞核大而规整,位于细胞中央。MCAO组小鼠的脑梗死区及周围组织中Fe2+和MDA含量较Sham组显著增加(P<0.001),MCAO+EXO组的Fe2+和MDA含量较MCAO组显著减少(P<0.01)。相较于Sham组,MCAO模型组小鼠的total GSH、GSSG、GSH含量均显著减少(P<0.01);相较于MCAO组,MCAO+EXO组小鼠的total GSH、GSH含量显著增加(P<0.001),GSSG含量无显著变化(P>0.05)。相较于Sham组,MCAO组小鼠的NRF2、SLC7A11、GPX4 mRNA和蛋白表达水平均显著降低(P<0.01,P<0.001);相较于MCAO组,MCAO+EXO组的NRF2、SLC7A11、GPX4 mRNA及蛋白表达水平均显著升高(P<0.05)。 结论 在小鼠MCAO模型中,尾静脉注射hAMSCs来源的外泌体可以改善小鼠运动功能,减少梗死面积,保护神经细胞形态,降低神经损伤程度。外泌体可能通过激活NRF2/SLC7A11/GPX4通路,减少MCAO模型小鼠的脑神经细胞铁死亡而发挥保护作用。
中图分类号:
徐英韬,王蒙蒙,林平,等. 外泌体通过NRF2/SLC7A11/GPX4通路调控铁死亡治疗小鼠缺血性脑卒中[J]. 实验动物与比较医学, 2026, 46(1): 20-31. DOI: 10.12300/j.issn.1674-5817.2025.034.
XU Yingtao,WANG Mengmeng,LIN Ping,et al. Exosomes Treat Ischemic Stroke by Regulation of Ferroptosis Through the NRF2/SLC7A11/GPX4 Pathway in Mice[J]. Laboratory Animal and Comparative Medicine, 2026, 46(1): 20-31. DOI: 10.12300/j.issn.1674-5817.2025.034.
引物名称 Primer name | 引物序列 (5'→3') Sequence (5'→3') | 产物大小/bp Product size/bp |
|---|---|---|
| GAPDH | F: AGGTCGGTGTGAACGGATTTG; R: GGGGTCGTTGATGGCAACA | 165 |
| NRF2 | F: CTTTAGTCAGCGACAGAAGGAC; R: AGGCATCTTGTTTGGGAATGTG | 219 |
| SLC7A11 | F: CACCGGGGTCGGTTTTCTTA; R: GGCAGATGGCCAAGCTTTTG | 200 |
| GPX4 | F: CTATGGTCCCATGGAGGAGC; R: AGGCAGACCTTCATGAGTGC | 185 |
表1 PCR引物序列
Table1 Sequence of primers used in PCR
引物名称 Primer name | 引物序列 (5'→3') Sequence (5'→3') | 产物大小/bp Product size/bp |
|---|---|---|
| GAPDH | F: AGGTCGGTGTGAACGGATTTG; R: GGGGTCGTTGATGGCAACA | 165 |
| NRF2 | F: CTTTAGTCAGCGACAGAAGGAC; R: AGGCATCTTGTTTGGGAATGTG | 219 |
| SLC7A11 | F: CACCGGGGTCGGTTTTCTTA; R: GGCAGATGGCCAAGCTTTTG | 200 |
| GPX4 | F: CTATGGTCCCATGGAGGAGC; R: AGGCAGACCTTCATGAGTGC | 185 |
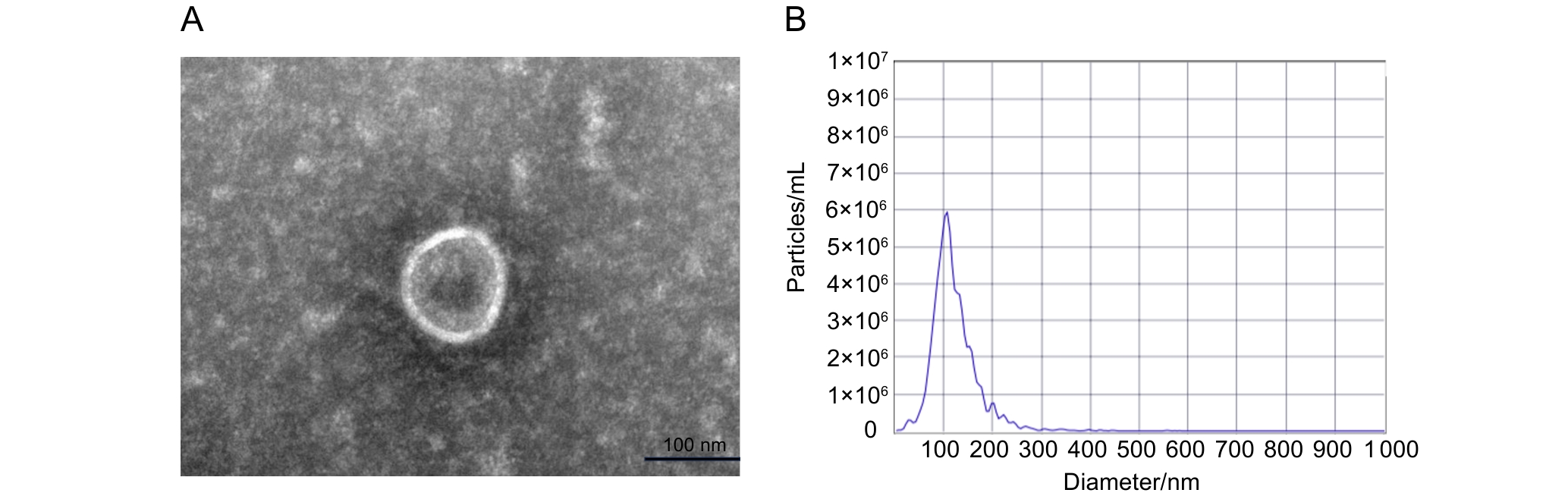
图1 外泌体的电镜观测和粒径分析
Figure 1 Electron microscopic observation and particle size analysis of exosomes
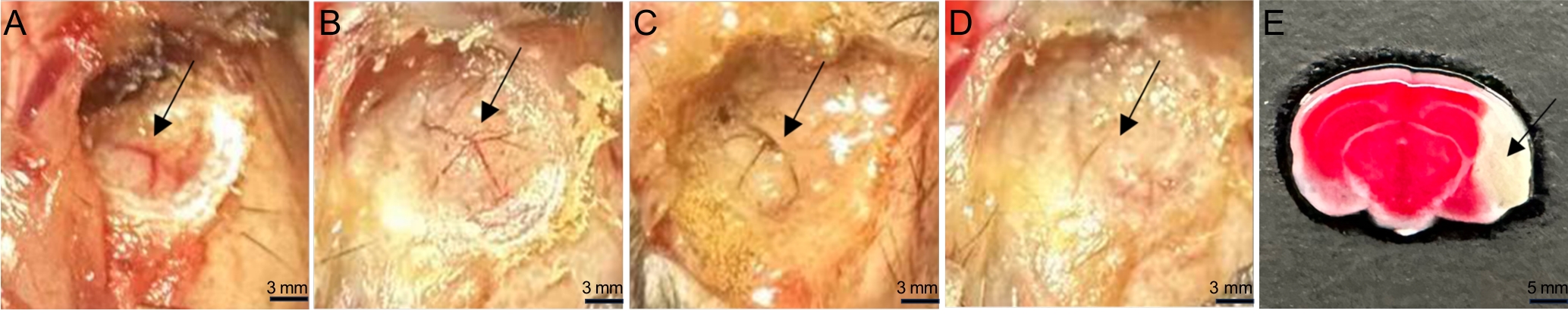
图2 MCAO小鼠模型建立过程中MCA血流变化和脑梗死区TTC染色表现
Figure 2 MCA blood flow changes during MCAO model establishment and TTC staining of the cerebral infarction area
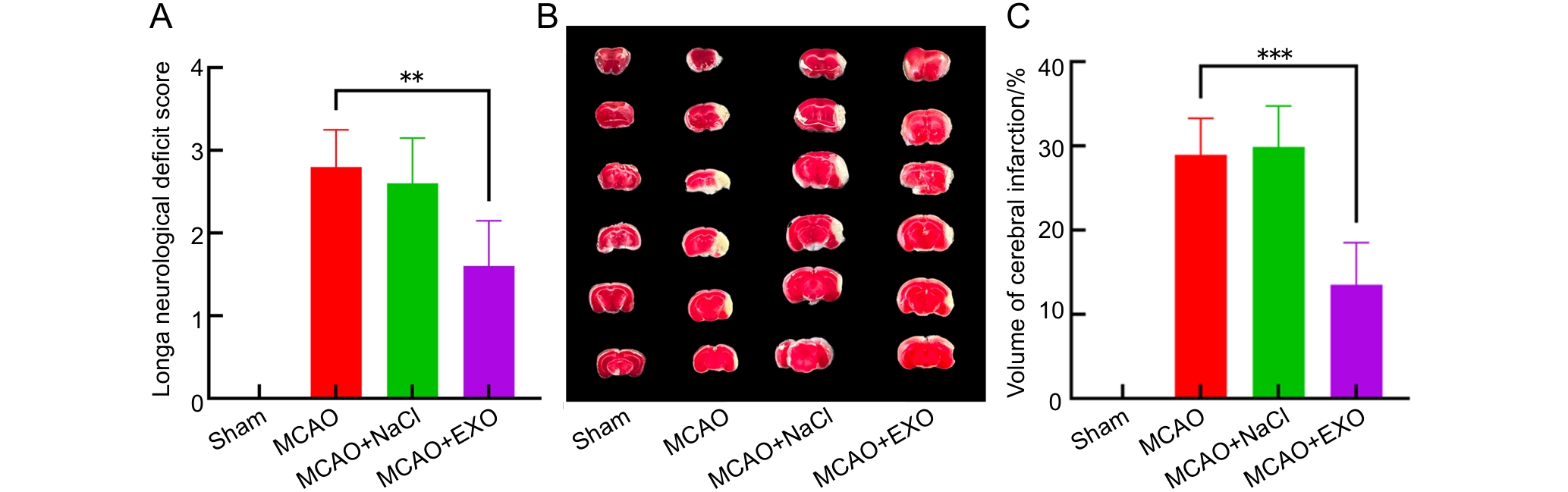
图3 各组小鼠神经功能缺损评分及脑梗死体积占比分析
注:A,各组小鼠神经功能Longa评分;B,各组小鼠脑切片的2,3,5-三苯基氯化四氮唑(TTC)染色结果,每列6张切片为1只小鼠的完整大脑,正常脑组织为红色,梗死区域脑组织为白色;C,各组小鼠脑梗死体积占比。假手术组(Sham)只暴露大脑中动脉,不电凝血管;模型组(MCAO)使用高频手术镊以8 W功率电凝大脑中动脉;模型+生理盐水组(MCAO+NaCl)先通过尾静脉注射100 μL生理盐水,然后进行电凝法造模;模型+外泌体组(MCAO+EXO)先通过尾静脉注射100 μL来自人羊膜间充质干细胞(hAMSCs)培养上清液的外泌体(9.5×1011个/mL),然后进行电凝法造模。每组小鼠各8只,**P<0.01,***P<0.001。
Figure 3 Analysis of neurological deficit scores and the percentages of cerebral infarction volume in each group of mice
Note:A, Neurological Longa scores of each group of mice; B, 2,3,5-triphenyltetrazolium chloride (TTC) staining results of brain sections in each group of mice, each column contains six slices representing the complete brain of one mouse, normal brain tissue was red, while brain tissue in the infarcted area was white; C, The percentages of brain infarction volume in each group of mice. The sham operation group (Sham) only exposed the middle cerebral artery without electrocoagulation of the vessel; the model group (MCAO) used high-frequency surgical forceps to electrocoagulate the middle cerebral artery at a power of 8 W; the model + normal saline group (MCAO+NaCl) first injected 100 μL of normal saline through the tail vein, and then performed electrocoagulation modeling; the model + exosome group (MCAO+EXO) first injected 100 μL of exosomes (9.5×1011 particles per mL) from the culture supernatant of human amniotic mesenchymal stem cells (hAMSCs) through the tail vein, and then performed electrocoagulation modeling. There were eight mice in each group. **P<0.01, ***P<0.001.
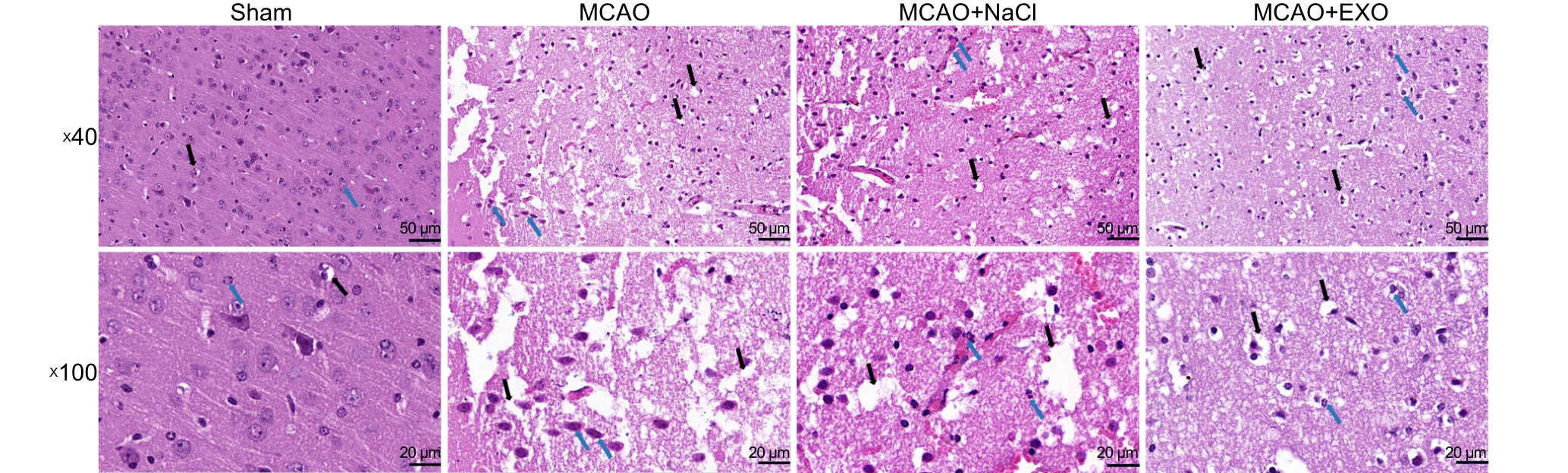
图4 各组小鼠脑组织切片HE染色观察神经细胞形态
Figure 4 HE staining of brain tissue sections from each group of mice to observe neuronal morphology
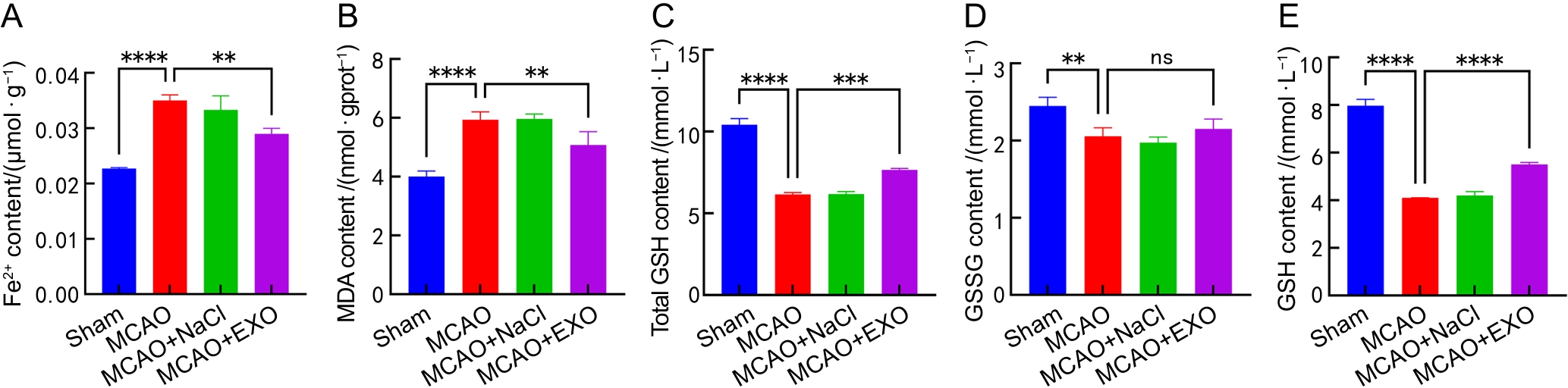
图5 各组小鼠脑组织中Fe2+、MDA、total GSH、GSSG和GSH含量的微量比色法分析结果
注:A~E,各组小鼠脑组织中亚铁离子(Fe2+)、丙二醛(MDA)、总谷胱甘肽(total GSH)、氧化型谷胱甘肽(GSSG)和还原型谷胱甘肽(GSH,即total GSH和GSSG含量的差值)含量分析。假手术组(Sham)只暴露大脑中动脉,不电凝血管;模型组(MCAO)使用高频手术镊以8 W功率电凝大脑中动脉;模型+生理盐水组(MCAO+NaCl)先通过尾静脉注射100 μL生理盐水,然后进行电凝法造模;模型+外泌体组(MCAO+EXO)先通过尾静脉注射100 μL来自人羊膜间充质干细胞(hAMSCs)培养上清液的外泌体(9.5×1011个/mL),然后进行电凝法造模。每组小鼠各8只,nsP >0.05,**P<0.01,***P<0.001,****P<0.000 1。
Figure 5 Microcolorimetric assays of Fe2+, MDA, total GSH, GSSG and GSH contents in the brains of each group of mice
Note:A-E, Contents of Fe2+, malondialdehyde (MDA), total glutathione (total GSH), oxidized glutathione (GSSG), and reduced glutathione (GSH) in the brains of mice from each group. Reduced GSH was calculated as the difference between the contents of total GSH and GSSG. The sham operation group (Sham) only exposed the middle cerebral artery without electrocoagulation of the vessel; the model group (MCAO) used high-frequency surgical forceps to electrocoagulate the middle cerebral artery at a power of 8 W; the model + normal saline group (MCAO+NaCl) first injected 100 μL of normal saline through the tail vein, and then performed electrocoagulation modeling; the model + exosome group (MCAO+EXO) first injected 100 μL of exosomes (9.5×1011 particles per mL) from the culture supernatant of human amniotic mesenchymal stem cells (hAMSCs) through the tail vein, and then performed electrocoagulation modeling. There were eight mice in each group. nsP >0.05, **P<0.01, ***P<0.001, ****P<0.000 1.
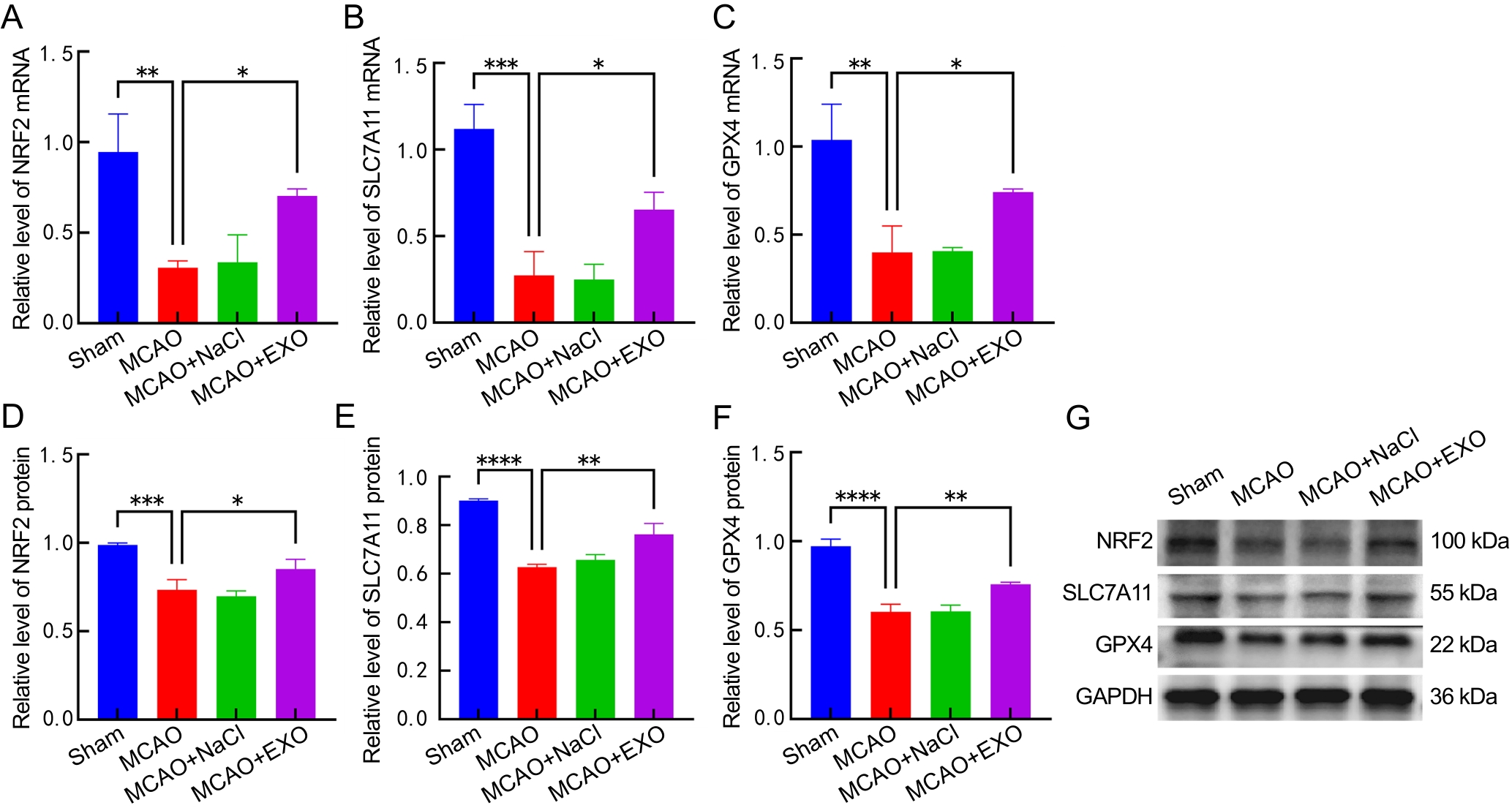
图6 各组小鼠脑组织中NRF2、SLC7A11、GPX4 mRNA及蛋白表达检测分析结果
注:A~C,各组小鼠脑组织中核因子-红细胞系2相关因子2(NRF2)、溶质载体家族7成员11(SLC7A11)和谷胱甘肽过氧化物酶4(GPX4)的mRNA转录水平分析;D~F,各组小鼠脑组织中NRF2、SLC7A11和GPX4蛋白相对表达量分析;G,各组小鼠脑组织中NRF2、SLC7A11、GPX4和GAPDH的蛋白质印迹结果图。假手术组(Sham)只暴露大脑中动脉,不电凝血管;模型组(MCAO)使用高频手术镊以8 W功率电凝大脑中动脉;模型+生理盐水组(MCAO+NaCl)先通过尾静脉注射100 μL生理盐水,然后进行电凝法造模;模型+外泌体组(MCAO+EXO)先通过尾静脉注射100 μL来自人羊膜间充质干细胞(hAMSCs)培养上清液的外泌体(9.5×1011个/mL),然后进行电凝法造模。每组小鼠各8只,*P<0.05,**P<0.01,***P<0.001,****P<0.000 1。
Figure 6 Detection and analysis of mRNA and protein expression of NRF2, SLC7A11 and GPX4 in the brains of each group of mice
Note:A-C, mRNA transcription levels of nuclear factor-erythroid 2-related factor 2 (NRF2), solute carrier family 7 member 11 (SLC7A11), and glutathione peroxidase 4 (GPX4) in the brains of mice from each group. D-F, Relative protein expression levels of NRF2, SLC7A11, and GPX4 in the brains of mice from each group. G, Western blotting images of NRF2, SLC7A11, GPX4, and GAPDH. The sham operation group (Sham) only exposed the middle cerebral artery without electrocoagulation of the vessel; the model group (MCAO) used high-frequency surgical forceps to electrocoagulate the middle cerebral artery at a power of 8 W; the model + normal saline group (MCAO+NaCl) first injected 100 μL of normal saline through the tail vein, and then performed electrocoagulation modeling; the model + exosome group (MCAO+EXO) first injected 100 μL of exosomes (9.5×1011 particles per mL) from the culture supernatant of human amniotic mesenchymal stem cells (hAMSCs) through the tail vein, and then performed electrocoagulation modeling. There were eight mice in each group. *P<0.05, **P<0.01, ***P<0.001, ****P<0.000 1.
| [1] | ZHU Z B, SHI M Y, YU Q, et al. Burden and risk factors of stroke worldwide and in China: An analysis from the Global Burden of Disease Study 2021[J]. Chin Med J (Engl), 2025,138(20):2588-2595. DOI: 10.1097/CM9.0000000000003778 . |
| [2] | GREEN T L, MCNAIR N D, HINKLE J L, et al. Care of the patient with acute ischemic stroke (posthyperacute and prehospital discharge): update to 2009 comprehensive nursing care scientific statement: a scientific statement from the American heart association[J]. Stroke, 2021, 52(5): e179-e197. DOI:10.1161/STR.0000000000000357 . |
| [3] | QIN C, YANG S, CHU Y H, et al. Signaling pathways involved in ischemic stroke: molecular mechanisms and therapeutic interventions[J]. Signal Transduct Target Ther, 2022, 7(1):215. DOI:10.1038/s41392-022-01064-1 . |
| [4] | TUO Q Z, LEI P. Ferroptosis in ischemic stroke: Animal models and mechanisms[J]. Zool Res, 2024, 45(6):1235-1248. DOI:10.24272/j.issn.2095-8137.2024.239 . |
| [5] | ZHANG Y F, LU X Y, TAI B, et al. Ferroptosis and its multifaceted roles in cerebral stroke[J]. Front Cell Neurosci, 2021, 15:615372. DOI: 10.3389/fncel.2021.615372 . |
| [6] | JIANG X J, STOCKWELL B R, CONRAD M. Ferroptosis: mechanisms, biology and role in disease[J]. Nat Rev Mol Cell Biol, 2021, 22(4):266-282. DOI:10.1038/s41580-020-00324-8 . |
| [7] | YANG Y P, NICOL C J B, CHIANG M C. A review of the neuroprotective properties of exosomes derived from stem cells and exosome-coated nanoparticles for treating neurodegenerative diseases and stroke[J]. Int J Mol Sci, 2025, 26(8):3915. DOI: 10.3390/ijms26083915 . |
| [8] | SONG Y, ZHANG T, SHI P, et al. Exosomes derived from human amniotic mesenchymal stem cells promotes angiogenesis in hUVECs by delivering novel miRNA N-194[J]. Mol Med, 2025, 31:173. DOI:10.1186/s10020-025-01192-8 . |
| [9] | SUN J, YUAN Q, GUO L,et al. Brain microvascular endothelial cell-derived exosomes protect neurons from ischemia-reperfusion injury in mice[J]. Pharmaceuticals (Basel), 2022, 15(10):1287. DOI:10.3390/ph15101287 . |
| [10] | QIN J, ZHOU L, YU L, et al. Exosomes derived from HUVECs alleviate ischemia-reperfusion induced inflammation in neural cells by upregulating KLF14 expression[J]. Front Pharmacol, 2024, 15:1365928. DOI: 10.3389/fphar.2024. 1365928 . |
| [11] | CHEN Y X, LI B F, QUAN J, et al. Inhibition of ferroptosis by mesenchymal stem cell-derived exosomes in acute spinal cord injury: role of Nrf2/GCH1/BH4 axis[J]. Neurospine, 2024, 21(2):642-655. DOI:10.14245/ns.2448038.019 . |
| [12] | LI X, ZHANG X, LIU Y J, et al. Exosomes derived from mesenchyml stem cells ameliorate oxygen-glucose deprivation/reoxygenation-induced neuronal injury via transferring microRNA-194 and targeting Bach1[J]. Tissue Cell, 2021, 73:101651. DOI:10.1016/j.tice.2021.101651 . |
| [13] | HUO S Z, SHI P, PANG X N. Culture and identification of human amniotic mesenchymal stem cells[J]. Chin Med Sci J, 25(4):211-214. DOI: 10.1016/s1001-9294(11)60004-7 . |
| [14] | WANG M M, BAI Y, CHI H T, et al. miR-451 protects against ischemic stroke by targeting Phd3[J]. Exp Neurol, 2021, 343:113777. DOI:10.1016/j.expneurol.2021.113777 . |
| [15] | LONGA E Z, WEINSTEIN P R, CARLSON S, et al. Reversible middle cerebral artery occlusion without craniectomy in rats[J]. Stroke, 1989, 20(1): 84-91. DOI: 10.1161/01.str.20.1.84 . |
| [16] | SWANSON R A, MORTON M T, TSAO-WU G, et al. A semiautomated method for measuring brain infarct volume[J]. J Cereb Blood Flow Metab, 1990, 10(2): 290-293. DOI:10.1038/jcbfm.1990.47 . |
| [17] | LI C, LUO Y P, LI S G, et al. Mechanistic insights of neuronal death and neuroprotective therapeutic approaches in stroke[J]. Neural Regen Res, 2025, 21(3):869-886. DOI:10.4103/NRR.NRR-D-24-01324 . |
| [18] | TANG D L, CHEN X, KANG R, et al. Ferroptosis: molecular mechanisms and health implications[J]. Cell Res, 2021, 31(2):107-125. DOI:10.1038/s41422-020-00441-1 . |
| [19] | SHEN K, WANG X J, WANG Y W, et al. miR-125b-5p in adipose derived stem cells exosome alleviates pulmonary microvascular endothelial cells ferroptosis via Keap1/Nrf2/GPX4 in sepsis lung injury[J]. Redox Biol, 2023, 62:102655. DOI:10.1016/j.redox.2023.102655 . |
| [20] | LIN F Y, CHEN W Y, ZHOU J H, et al. Mesenchymal stem cells protect against ferroptosis via exosome-mediated stabilization of SLC7A11 in acute liver injury[J]. Cell Death Dis, 2022, 13(3):271. DOI:10.1038/s41419-022-04708-w . |
| [21] | 邢凤英, 周颖, 马政文, 等. 线栓法大鼠大脑中动脉脑缺血模型的评价方法初探[J]. 实验动物与比较医学, 2013, 33(5): 339-346. DOI: 10.3969/j.issn.1674-5817.2013.05.003 . |
| XING F Y, ZHOU Y, MA Z W, et al. Preliminary study for evaluation system of rat permanent cerebral artery occlusion model by intraluminal suture method[J]. Lab Anim Comp Med, 2013, 33(5): 339-346. DOI: 10.3969/j.issn.1674-5817.2013.05.003 . | |
| [22] | SUN Y, YANG X, XU L, et al. The role of Nrf2 in relieving cerebral ischemia-reperfusion injury[J]. Curr Neuropharmacol, 2023, 21(6):1405-1420. DOI: 10.2174/1570159X21666221129100308 . |
| [23] | VILLAVICENCIO-TEJO F, OLESEN M A, ARÁNGUIZ A, et al. Activation of the Nrf2 pathway prevents mitochondrial dysfunction induced by caspase-3 cleaved Tau: implications for Alzheimer's disease[J]. Antioxidants (Basel), 2022,11(3):515. DOI: 10.3390/antiox11030515 . |
| [24] | LIU H, ZHANG T A, ZHANG W Y, et al. Rhein attenuates cerebral ischemia-reperfusion injury via inhibition of ferroptosis through NRF2/SLC7A11/GPX4 pathway[J]. Exp Neurol, 2023, 369:114541. DOI:10.1016/j.expneurol.2023.114541 . |
| [25] | FU C, WU Y F, LIU S J, et al. Rehmannioside A improves cognitive impairment and alleviates ferroptosis via activating PI3K/AKT/Nrf2 and SLC7A11/GPX4 signaling pathway after ischemia[J]. J Ethnopharmacol, 2022, 289:115021. DOI:10.1016/j.jep.2022.115021 . |
| [1] | 杨韵蓉, 吴文郁, 谭跃, 严国锋, 李垚, 卢今. 脑卒中动物模型构建与评价方法评述[J]. 实验动物与比较医学, 2026, 46(1): 94-106. |
| [2] | 张一粟, 刘欣茹, 武若杰, 刘睿, 欧阳红, 李晓红. 慢性不可预知性应激与完全弗氏佐剂、福尔马林诱导的妊娠期疼痛-抑郁共病小鼠模型的构建与评价[J]. 实验动物与比较医学, 2024, 44(3): 259-269. |
| [3] | 刘佳, 叶岩荣, 沈赟, 唐启瑛, 陈梅卿, 易可慧, 陈少壮. 银杏内酯B通过调控脑内T细胞特性及与胶质细胞间相互作用促进缺血性脑卒中小鼠的神经功能恢复[J]. 实验动物与比较医学, 2024, 44(2): 139-148. |
| [4] | 梁敏, 郭洋, 王津津, 朱梦妍, 池骏, 陈艳娟, 王成稷, 喻智澜, 沈如凌. Dmd基因突变小鼠构建及在肌肉及免疫系统的表型验证[J]. 实验动物与比较医学, 2024, 44(1): 42-51. |
| [5] | 林金杏, 王新栋, 白雪兵, 冯丽萍, 谢淑武, 陈秋生. 成年斑马鱼体肾的微细结构及其外泌体的分布鉴定[J]. 实验动物与比较医学, 2023, 43(5): 531-540. |
| [6] | 李晗, 张笑瑞, 张成芳. 间歇禁食法在改善奥氮平诱导小鼠代谢紊乱中的机制研究[J]. 实验动物与比较医学, 2023, 43(1): 3-10. |
| [7] | 朱彦兵, 白帆, 陶少鑫, 潘雨花蕾, 王欢, 赵羽商, 王崧, 于艳. 抑制磷脂酶D1活性可促进缺血性脑卒中模型小鼠神经功能恢复[J]. 实验动物与比较医学, 2022, 42(4): 322-332. |
| [8] | 董波, 刘嘉欣, 熊伟, 唐宋琪, 黄巍. 缺血性脑卒中动物模型的研究进展[J]. 实验动物与比较医学, 2022, 42(1): 54-61. |
| [9] | 李自发, 张浩, 任萌, 徐凯勇, 胡明会, 周苗苗, 王可洲. 槲皮素对镉致小鼠肝脏脂质代谢紊乱模型的保护效应[J]. 实验动物与比较医学, 2021, 41(4): 305-312. |
| [10] | 冯延, 武文卿, 张静远, 李煜, 李壘辰, 袁征, 崔淑芳. 裸鼹鼠皮肤成纤维细胞外泌体的分离及鉴定[J]. 实验动物与比较医学, 2020, 40(6): 506-512. |
| [11] | 洪胜辉, 张旭亮, 王芊芊, 刘 平, 刘迪文. 抑制素基因敲除小鼠模型的构建及表型初步分析[J]. 实验动物与比较医学, 2020, 40(4): 306-. |
| [12] | 李雷斌, 许 佳, 方远书, 姜正前, 裘颖儿, 王文倩, 赵先哲. 基于自发糖尿病模型小鼠代谢组学的全植物蛋白配方饲料研究#br#[J]. 实验动物与比较医学, 2020, 40(3): 204-. |
| [13] | 雷山, 刘强, 黄维金, 王佑春. 小鼠品系、性别和被毛对可视化假病毒感染模型的影响[J]. 实验动物与比较医学, 2019, 39(6): 423-428. |
| [14] | 牛博文, 陈丽香, 朱孟敏, 彭秀华, 秦波音, 李峰. C57BL/6J背景基因修饰小鼠精子冷冻保种及品系恢复方法[J]. 实验动物与比较医学, 2019, 39(2): 146-150. |
| [15] | 杨华, 赵亚娟, 欧强. 非酒精性脂肪性肝纤维化小鼠模型的建立及炎症因子的表达[J]. 实验动物与比较医学, 2017, 37(1): 20-24. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||