













实验动物与比较医学 ›› 2026, Vol. 46 ›› Issue (2): 178-190.DOI: 10.12300/j.issn.1674-5817.2025.175
荣文双1,2,3, 牛源菲4, 刘美婷2,3, 杨梦园5, 崔爽2,3, 马丽娜6, 富尧1, 王连嵋2,3( )(
)( ), 曹俊岭1,6(
), 曹俊岭1,6( )(
)( )
)
收稿日期:2025-10-24
修回日期:2026-02-05
出版日期:2026-04-25
发布日期:2026-04-20
通讯作者:
作者简介:荣文双(1996—),女,硕士研究生,研究方向:中医优势病种的中医药治疗。E-mail: rongwenshuang123@163.com
基金资助:
RONG Wenshuang1,2,3, NIU Yuanfei4, LIU Meiting2,3, YANG Mengyuan5, CUI Shuang2,3, MA Lina6, FU Yao1, WANG Lianmei2,3( )(
)( ), CAO Junling1,6(
), CAO Junling1,6( )(
)( )
)
Received:2025-10-24
Revised:2026-02-05
Published:2026-04-25
Online:2026-04-20
Contact:
CAO Junling (ORCID: 0000-0001-7640-8713), E-mail: caojunling72@163.com;摘要:
目的 观察比较颌下腺抗原和唾液腺抗原用于诱导干燥综合征(Sj?gren syndrome,SS)小鼠模型的建模效果,并与自发性SS模型NOD/LtJ小鼠进行比较,明确两种腺体抗原诱导的SS小鼠模型的表型与免疫特征。 方法 选用成年C57BL/6J小鼠(雌雄各半),分别采用颌下腺抗原和唾液腺抗原联合弗氏佐剂免疫小鼠,诱导建立SS模型;以PBS联合弗氏佐剂免疫小鼠作为对照。在第1天和第7天时采用抗原联合弗氏完全佐剂(Freund’s complete adjuvant,FCA)经小鼠背部皮下多点注射进行免疫诱导,第14天采用抗原联合弗氏不完全佐剂(Freund’s incomplete adjuvant,FIA)经小鼠背部皮下多点注射进行加强免疫诱导。同时设置雌性NOD/LtJ小鼠为自发性SS模型组,以其背景品系ICR小鼠为对照,进行对比分析。实验周期为4周,动态监测小鼠体重、饮水量及唾液流量的变化。在实验终点时收集小鼠各脏器组织与血清样本,称量颌下腺、胸腺和脾脏重量,计算脏器指数;对颌下腺和脾脏组织进行病理形态学分析;采用ELISA法检测血清中白细胞介素-17(interleukin-17,IL-17)含量;采用实时荧光定量PCR法检测颌下腺组织中SS抗原A(SS type A,SSA)和SS抗原B(SS type B,SSB)的mRNA表达水平。 结果 与雌性的PBS联合弗氏佐剂对照组相比,颌下腺抗原雌性组小鼠的饮水量明显增加(P<0.05),唾液分泌量明显减少(P<0.05),颌下腺指数、胸腺指数和脾脏指数均无明显差异(P>0.05),颌下腺出现灶性淋巴细胞浸润、脾脏边缘区扩大,血清IL-17水平明显升高(P<0.05),颌下腺组织中SSA/SSB表达水平无明显差异(P>0.05)。与雌性的PBS联合弗氏佐剂对照组相比,唾液腺抗原雌性组小鼠饮水量、唾液分泌量、颌下腺指数以及脾脏指数均无明显差异(P>0.05),但胸腺指数明显降低(P<0.01);颌下腺出现轻微炎性细胞浸润和腺体萎缩,脾脏白髓及边缘区略有扩大;血清IL-17水平与颌下腺组织中SSB表达水平均明显升高(P<0.01),颌下腺组织中SSA表达水平无明显变化(P>0.05)。与雄性的PBS联合弗氏佐剂对照组相比,除颌下腺抗原雄性组小鼠出现轻微颌下腺腺体萎缩外,各造模组的其余成模指标均没有明显差异(P>0.05)。与ICR对照组相比,NOD/LtJ模型组小鼠饮水量明显增加(P<0.05),唾液流量明显减少(P<0.01),颌下腺指数和脾脏指数均无明显差异(P>0.05),但胸腺指数明显增大(P<0.05);颌下腺病理灶性浸润明显,脾脏边缘区明显扩大,血清IL-17水平和颌下腺SSA/SSB表达水平均明显升高(P<0.05)。 结论 颌下腺抗原和唾液腺抗原均能诱导雌性C57BL/6J小鼠表现出SS相关特征,而颌下腺抗原诱导的SS相关表型较唾液腺抗原诱导更为明显,但弱于自发性雌性NOD/LtJ小鼠。用颌下腺抗原和唾液腺抗原接种雄性C57BL/6J小鼠未能诱导出明显的SS表型。
中图分类号:
荣文双,牛源菲,刘美婷,等. 抗原类型对诱导型干燥综合征小鼠建模的影响[J]. 实验动物与比较医学, 2026, 46(2): 178-190. DOI: 10.12300/j.issn.1674-5817.2025.175.
RONG Wenshuang,NIU Yuanfei,LIU Meiting,et al. Influence of Antigen Type on the Establishment of an Induced Sjögren Syndrome Mouse Model[J]. Laboratory Animal and Comparative Medicine, 2026, 46(2): 178-190. DOI: 10.12300/j.issn.1674-5817.2025.175.
引物名称 Primer name | 引物序列 Primer sequence |
|---|---|
| m-SSA-F | 5'-GCTGGTCTCACAAAGATCTCCTCC-3' |
| m-SSA-R | 5'-TCTCAGCCTCCACAGAAAGCG-3' |
| m-SSB-F | 5'-ATGACTGCTTTGGAGGCCAAAATC-3' |
| m-SSB-R | 5'-TGTCAGCCGGTTTAGCCTGTTGAA-3' |
| m-β-Actin-F | 5'-AGAGGGAAATCGTGCGTGAC-3' |
| m-β-Actin-R | 5'-CAATAGTGATGACCTGGCCGT-3' |
表1 PCR 引物序列
Table 1 PCR primer sequences
引物名称 Primer name | 引物序列 Primer sequence |
|---|---|
| m-SSA-F | 5'-GCTGGTCTCACAAAGATCTCCTCC-3' |
| m-SSA-R | 5'-TCTCAGCCTCCACAGAAAGCG-3' |
| m-SSB-F | 5'-ATGACTGCTTTGGAGGCCAAAATC-3' |
| m-SSB-R | 5'-TGTCAGCCGGTTTAGCCTGTTGAA-3' |
| m-β-Actin-F | 5'-AGAGGGAAATCGTGCGTGAC-3' |
| m-β-Actin-R | 5'-CAATAGTGATGACCTGGCCGT-3' |
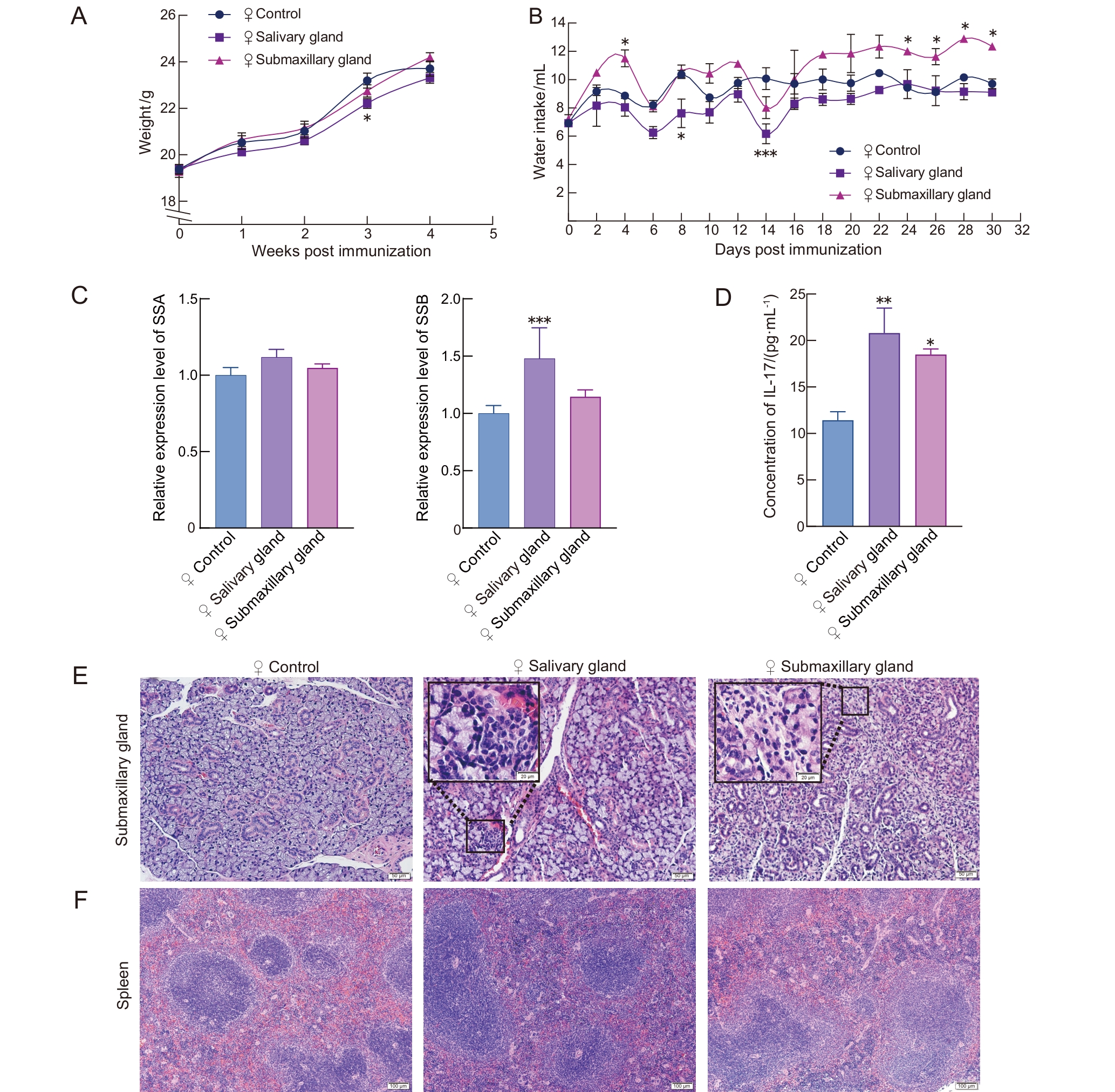
图 1 免疫诱导后雌性C57BL/6J小鼠的干燥综合征相关表型
Figure 1 Sj?gren syndrome-related phenotypes in antigen-induced female C57BL/6J mice
组别 Group | 第0天唾液流量/ (mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/ (mg·min-1) Day 30 salivary flow rate /(mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
PBS-佐剂雌性对照组 PBS-adjuvanted female control group | 6.60±1.11 | 14.77±1.04 | 0.28±0.03 | 0.26±0.04 | 0.44±0.04 |
唾液腺抗原雌性组 Salivary gland antigen female group | 6.28±1.10 | 13.10±1.05 | 0.29±0.03 | 0.20±0.02∗∗ | 0.51±0.13 |
颌下腺抗原雌性组 Submaxillary gland antigen female group | 6.14±1.16 | 10.52±1.06∗ | 0.30±0.03 | 0.24±0.04 | 0.50±0.06 |
表2 雌性C57BL/6J各组小鼠唾液流量和脏器指数 (
Table 2 Salivary flow rate and organ indices in female C57BL/6J mice of each group
组别 Group | 第0天唾液流量/ (mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/ (mg·min-1) Day 30 salivary flow rate /(mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
PBS-佐剂雌性对照组 PBS-adjuvanted female control group | 6.60±1.11 | 14.77±1.04 | 0.28±0.03 | 0.26±0.04 | 0.44±0.04 |
唾液腺抗原雌性组 Salivary gland antigen female group | 6.28±1.10 | 13.10±1.05 | 0.29±0.03 | 0.20±0.02∗∗ | 0.51±0.13 |
颌下腺抗原雌性组 Submaxillary gland antigen female group | 6.14±1.16 | 10.52±1.06∗ | 0.30±0.03 | 0.24±0.04 | 0.50±0.06 |
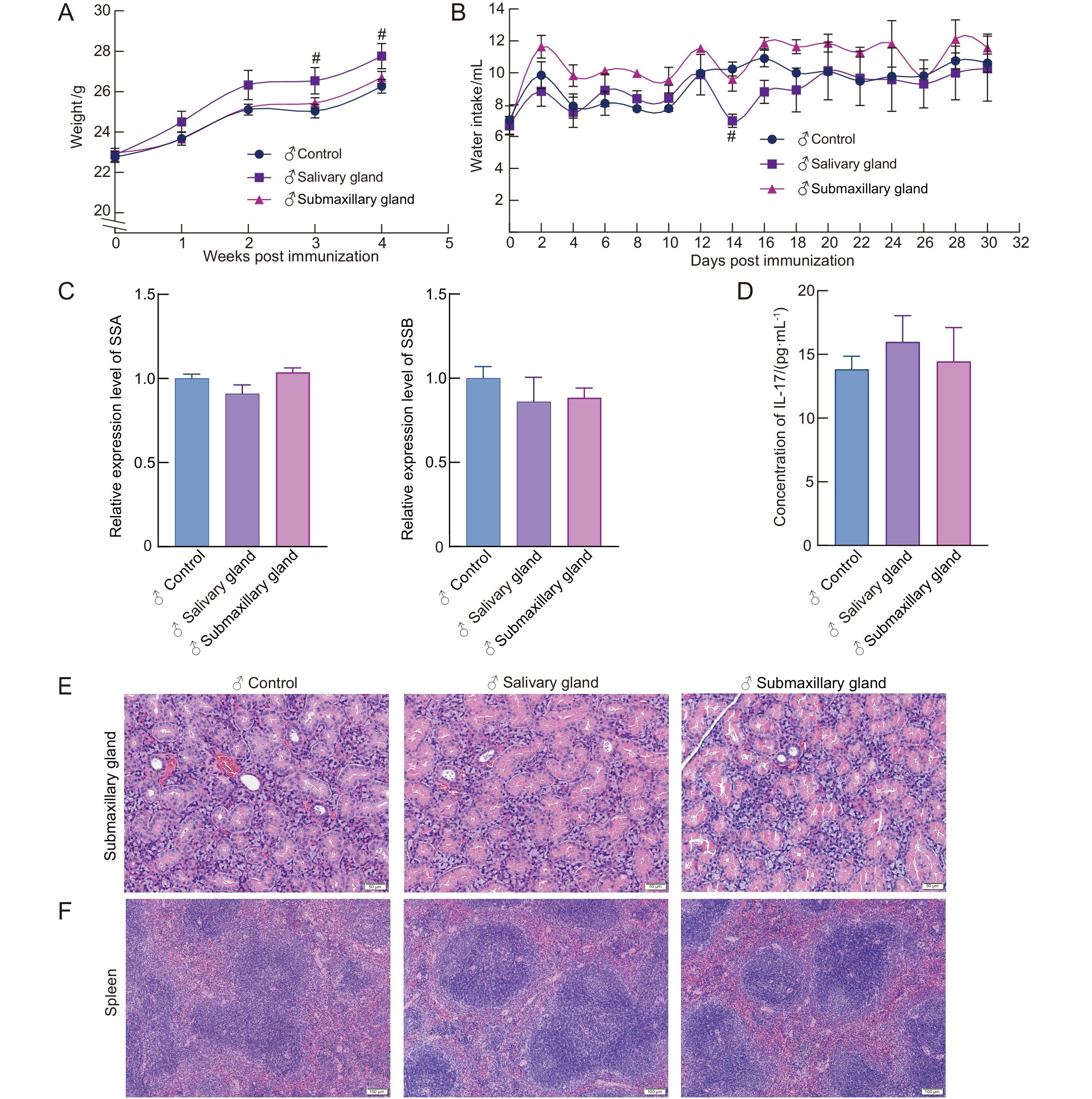
图 2 免疫诱导后雄性C57BL/6J小鼠的干燥综合征相关表型
Figure 2 Sj?gren syndrome-related phenotypes in antigen-induced male C57BL/6J mice
组别 Group | 第0天唾液流量/ (mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/(mg·min-1) Day 30 salivary flow rate /(mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
PBS-佐剂雄性对照组 PBS-adjuvanted male control group | 6.21±0.88 | 14.60±0.90 | 0.38±0.03 | 0.18±0.06 | 0.41±0.07 |
唾液腺抗原雄性组 Salivary gland antigen male group | 6.72±0.73 | 11.17±0.62 | 0.40±0.04 | 0.18±0.05 | 0.37±0.14 |
颌下腺抗原雄性组 Submaxillary gland antigen male group | 6.17±0.90 | 11.54±0.70 | 0.38±0.06 | 0.17±0.03 | 0.34±0.05 |
表3 雄性C57BL/6J各组小鼠的唾液流量和脏器指数 (
Table 3 Salivary flow rate and organ indices in male C57BL/6J mice of each group
组别 Group | 第0天唾液流量/ (mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/(mg·min-1) Day 30 salivary flow rate /(mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
PBS-佐剂雄性对照组 PBS-adjuvanted male control group | 6.21±0.88 | 14.60±0.90 | 0.38±0.03 | 0.18±0.06 | 0.41±0.07 |
唾液腺抗原雄性组 Salivary gland antigen male group | 6.72±0.73 | 11.17±0.62 | 0.40±0.04 | 0.18±0.05 | 0.37±0.14 |
颌下腺抗原雄性组 Submaxillary gland antigen male group | 6.17±0.90 | 11.54±0.70 | 0.38±0.06 | 0.17±0.03 | 0.34±0.05 |
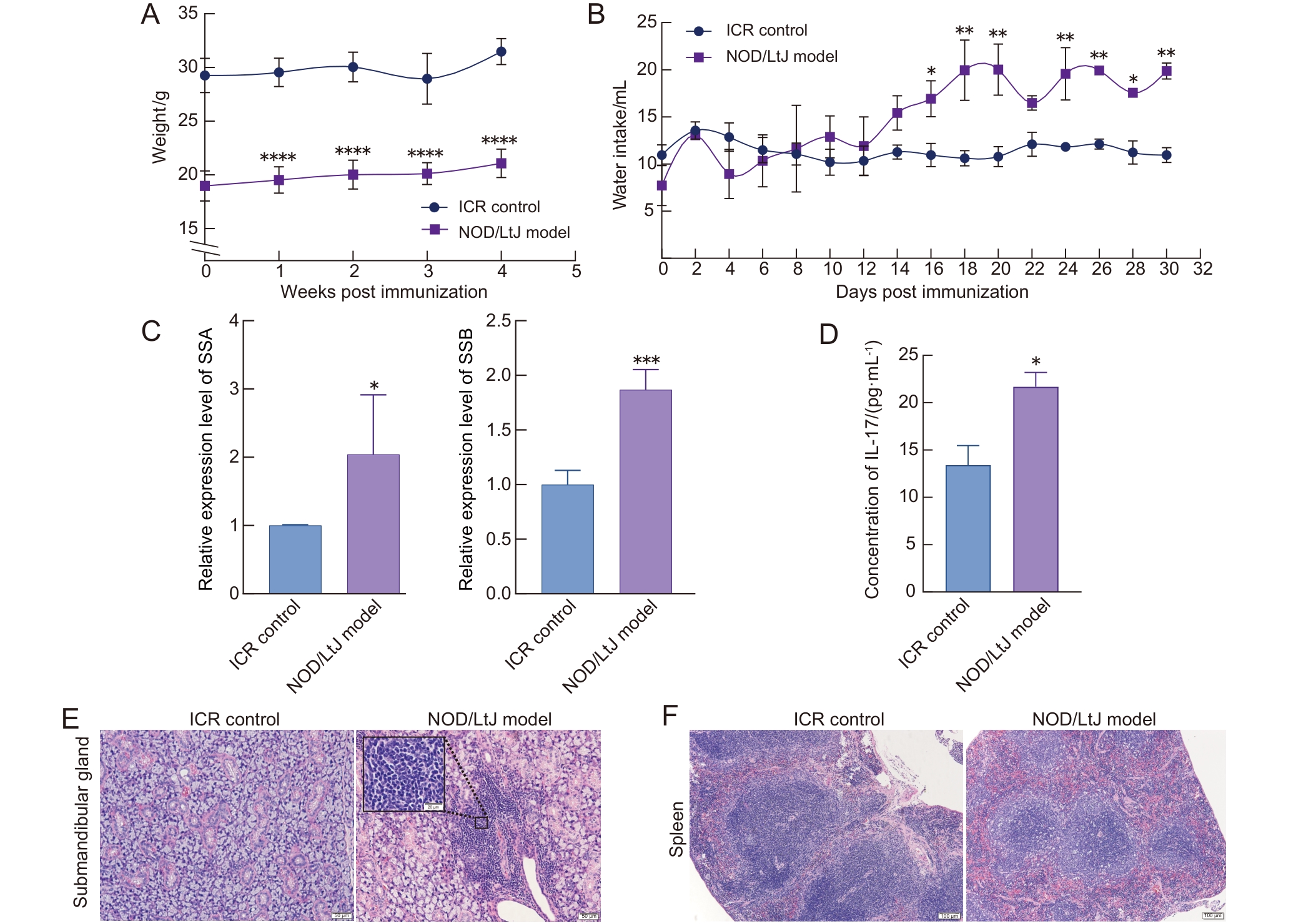
图 3 雌性NOD/LtJ模型小鼠与 ICR小鼠的干燥综合征相关表型
Figure 3 Sj?gren syndrome-related phenotypes in female NOD/LtJ model mice and ICR mice
组别 Group | 第0天唾液流量/(mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/(mg·min-1) Day 30 salivary flow rate / (mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
| ICR mice | 8.37±0.46 | 15.85±1.25 | 0.39±0.01 | 0.13±0.03 | 0.39±0.08 |
| NOD/LtJ mice | 6.42±0.05∗ | 5.11±0.46∗∗ | 0.40±0.04 | 0.16±0.03∗ | 0.31±0.05 |
表4 雌性NOD/LtJ模型小鼠与ICR小鼠的唾液流量和脏器指数比较 (
Table 4 Salivary flow rate and organ indices in female NOD/LtJ model mice and ICR mice
组别 Group | 第0天唾液流量/(mg·min-1) Day 0 salivary flow rate /(mg·min-1) | 第30天唾液流量/(mg·min-1) Day 30 salivary flow rate / (mg·min-1) | 颌下腺指数 Submaxillary gland index | 胸腺指数 Thymus index | 脾指数 Spleen index |
|---|---|---|---|---|---|
| ICR mice | 8.37±0.46 | 15.85±1.25 | 0.39±0.01 | 0.13±0.03 | 0.39±0.08 |
| NOD/LtJ mice | 6.42±0.05∗ | 5.11±0.46∗∗ | 0.40±0.04 | 0.16±0.03∗ | 0.31±0.05 |
| [1] | 刘维, 何东仪, 张文, 等. 干燥综合征中西医结合诊疗指南[J]. 中草药, 2025, 56(15): 5333-5346. DOI: 10.7501/j.issn.0253-2670.2025.15.001 . |
| LIU W, HE D Y, ZHANG W, et al. Guideline for diagnosis and treatment of Sjögren's syndrome with integrated traditional Chinese and Western medicine[J]. Chin Tradit Herb Drugs, 2025, 56(15): 5333-5346. DOI: 10.7501/j.issn.0253-2670.2025.15.001 . | |
| [2] | 张欢, 刘春红, 吴斌. 原发性干燥综合征的流行病学研究进展[J]. 现代预防医学, 2020, 47(16): 3056-3058, 3063. DOI: 10.20043/j.cnki.mpm.2020.16.041 . |
| ZHANG H, LIU C H, WU B. Advances in the epidemiology of primary Sjögren's syndrome[J]. Mod Prev Med, 2020, 47(16): 3056-3058, 3063. DOI: 10.20043/j.cnki.mpm.2020.16.041 . | |
| [3] | FAIRWEATHER D, BEETLER D J, MCCABE E J, et al. Mechanisms underlying sex differences in autoimmunity[J]. J Clin Investig, 2024, 134(18): e180076. DOI: 10.1172/jci180076 . |
| [4] | GAO Y Z, CHEN Y, ZHANG Z J, et al. Recent advances in mouse models of Sjögren's syndrome[J]. Front Immunol, 2020, 11: 1158. DOI: 10.3389/fimmu.2020.01158 . |
| [5] | JIANG T T, LIU X Q, WANG S M, et al. Paeoniflorin alleviated experimental Sjögren's syndrome by inhibiting NLRP3 inflammasome activation of submandibular gland cells via activating Nrf2/HO-1 pathway[J]. Free Radic Biol Med, 2025, 233: 355-364. DOI: 10.1016/j.freeradbiomed.2025.03.043 . |
| [6] | LIN X, RUI K, DENG J, et al. Th17 cells play a critical role in the development of experimental Sjögren's syndrome[J]. Ann Rheum Dis, 2015, 74(6): 1302-1310. DOI: 10.1136/annrheumdis-2013-204584 . |
| [7] | BAGAVANT H, DURSLEWICZ J, PYCLIK M, et al. Age-associated B cell infiltration in salivary glands represents a hallmark of Sjögren's-like disease in aging mice[J]. Geroscience, 2024, 46(6): 6085-6099. DOI: 10.1007/s11357-024-01159-3 . |
| [8] | XU J J, LIU O S, WANG D D, et al. In vivo generation of SSA/ro antigen-specific regulatory T cells improves experimental Sjögren's syndrome in mice[J]. Arthritis Rheumatol, 2022, 74(10): 1699-1705. DOI: 10.1002/art.42244 . |
| [9] | ARVIDSSON G, CZARNEWSKI P, JOHANSSON A, et al. Multimodal single-cell sequencing of B cells in primary Sjögren's syndrome[J]. Arthritis Rheumatol, 2024, 76(2): 255-267. DOI: 10.1002/art.42683 . |
| [10] | LEE A Y S, PUTTY T, LIN M W, et al. Isolated anti-Ro52 identifies a severe subset of Sjögren's syndrome patients[J]. Front Immunol, 2023, 14: 1115548. DOI: 10.3389/fimmu.2023. 1115548 . |
| [11] | WU Y, PENG L, FENG P Y, et al. Gut microbes consume host energy and reciprocally provide beneficial factors to sustain a symbiotic relationship with the host[J]. Sci Total Environ, 2023, 904: 166773. DOI: 10.1016/j.scitotenv.2023.166773 . |
| [12] | LUO Y, ZENG L T, WANG Y N, et al. Artemisinin derivatives modulate KEAP1-NRF2-xCT pathway to alleviate Sjögren's disease: insights from scRNA-seq and systems biology[J]. Front Immunol, 2025, 16: 1626230. DOI: 10.3389/fimmu. 2025. 1626230 . |
| [13] | 贵州省中西医结合学会. 2016ACR/EULAR共识: 原发性干燥综合征的最新分类标准[C/OL]//2019年贵州省中医、中西医结合风湿病学术会议论文集. (2019-04-26)[2025-10-23]. . |
| Guizhou Provincial Association of Integrated Traditional Chinese and Western Medicine. 2016ACR/EULAR consensus: updated classification criteria for primary Sjögren's syndrome[C/OL]//Proceedings of the 2019 Guizhou Provincial Conference on Rheumatology in Traditional Chinese Medicine and Integrated Traditional and Western Medicine. (2019-04-26)[2025-10-23]. . | |
| [14] | VAN GINKEL M S, NAKSHBANDI U, ARENDS S, et al. Increased diagnostic accuracy of the labial gland biopsy in primary Sjögren syndrome when multiple histopathological features are included[J]. Arthritis Rheumatol, 2024, 76(3): 421-428. DOI: 10.1002/art.42723 . |
| [15] | MATSUI K, SANO H. T helper 17 cells in primary Sjögren's syndrome[J]. J Clin Med, 2017, 6(7): 65. DOI: 10.3390/jcm 6070065 . |
| [16] | ZHAN Q P, ZHANG J N, LIN Y B, et al. Pathogenesis and treatment of Sjogren's syndrome: Review and update[J]. Front Immunol, 2023, 14: 1127417. DOI: 10.3389/fimmu. 2023. 1127417 . |
| [17] | FLOREZI G P, BARONE F P, PELISSARI C, et al. Salivary Th17-associated cytokines as potential biomarkers in primary Sjögren's disease[J]. Oral Surg Oral Med Oral Pathol Oral Radiol, 2025, 140(4): 428-435. DOI: 10.1016/j.oooo.2025.04.171 . |
| [18] | 郭朝焱. 病理诊断要点与应用[M]. 南昌: 江西科学技术出版社, 2023. |
| GUO C Y. Key points and applications of pathological diagnosis[M]. Nanchang: Jiangxi Science and Technology Press, 2023. | |
| [19] | YAO Y, MA J F, CHANG C, et al. Immunobiology of T cells in Sjögren's syndrome[J]. Clin Rev Allergy Immunol, 2021, 60(1): 111-131. DOI: 10.1007/s12016-020-08793-7 . |
| [20] | ROBERT P A, ARULRAJ T, MEYER-HERMANN M. Germinal centers are permissive to subdominant antibody responses[J]. Front Immunol, 2024, 14: 1238046. DOI: 10.3389/fimmu. 2023.1238046 . |
| [21] | PASUPULETI D, BAGWE P, FERGUSON A, et al. Evaluating nanoparticulate vaccine formulations for effective antigen presentation and T-cell proliferation using an in vitro overlay assay[J]. Vaccines, 2024, 12(9): 1049. DOI: 10.3390/vaccines 12091049 . |
| [22] | TURNER J D, TSAPPARELLI J, GILLIGAN L C, et al. Hormonal risk factors and androgen and glucocorticoid dysregulation in Sjogren's disease and non-Sjogren's sicca[J]. Rheumatology, 2026, 65: keaf546. DOI: 10.1093/rheumatology/keaf546 . |
| [23] | CHATZIS L G, GOULES A V, TZIOUFAS A G. Searching for the ″X factor″ in Sjögren's syndrome female predilection[J]. Clin Exp Rheumatol, 2021, 39(6): 206-214. DOI: 10.55563/clinexprheumatol/88dyrn . |
| [24] | 陈冬志, 赵会娟, 尹晓琳, 等. NOD/LtJ小鼠Ⅰ型糖尿病不同发病阶段细胞免疫状态研究[J]. 中国免疫学杂志, 2019, 35(5): 526-533. DOI: 10.3969/j.issn.1000-484X.2019.05.003 . |
| CHEN D Z, ZHAO H J, YIN X L, et al. Study on cellular immune status of NOD/LtJ mice at different stages of typeⅠdiabetes[J]. Chin J Immunol, 2019, 35(5): 526-533. DOI: 10.3969/j.issn.1000-484X.2019.05.003 . | |
| [25] | WONG F S, PEARSON J A, WEN L. Is the NOD mouse a good model for type 1 diabetes?[J]. Diabetologia, 2026, 69(1): 3-19. DOI: 10.1007/s00125-025-06579-0 . |
| [1] | 宋静, 杨宗统, 李晓晶, 李自发, 苏凤云, 徐东川, 隋在云. 泻白散对过敏性哮喘大鼠肺、肠组织形态结构及PI3K和Akt表达水平的影响[J]. 实验动物与比较医学, 2026, 46(2): 191-204. |
| [2] | 孙强. 有关仿制药质量监管缺位与FDA新政的思考[J]. 实验动物与比较医学, 2026, 46(2): 306-310. |
| [3] | 赵赫, 张涛, 李丽, 肖宇宙, 安学芳, 张帆. 猪抑制素多克隆抗体对C57BL/6J小鼠超数排卵的增效作用[J]. 实验动物与比较医学, 2026, 46(2): 271-278. |
| [4] | 赵赫, 张涛, 肖宇宙, 李丽, 安学芳, 张帆. 动物生物安全二级实验室猫感染实验的生物风险控制[J]. 实验动物与比较医学, 2026, 46(2): 242-250. |
| [5] | 王晨, 李学波, 贺东华, 常亮堂. 实验狨猴信息管理系统的设计与应用[J]. 实验动物与比较医学, 2026, 46(2): 231-241. |
| [6] | 王莹, 纪文韬, 徐少琼, 陈国元, 冯洁, 吴宝金. 实验动物屏障设施环境微生物污染情况的监测与分析[J]. 实验动物与比较医学, 2026, 46(2): 222-230. |
| [7] | 刘赛, 付彬, 李思迪, 陈志达, 张悦, 郭中坤, 王永安, 王可洲. Adra2a通过MAPK信号通路介导LPS诱导的Lbp-/- 小鼠肝细胞炎性反应[J]. 实验动物与比较医学, 2026, 46(2): 212-221. |
| [8] | 崔畅婉, 路一平, 于淼, 王爽, 吴思, 孙峥嵘. 溶质载体家族7成员5抑制剂JPH203对单侧输尿管梗阻诱导小鼠肾纤维化的抑制作用[J]. 实验动物与比较医学, 2026, 46(2): 205-211. |
| [9] | 吴宪文, 刘丽丽, 陈烨, 徐国恒. 繁育体系中育成期小鼠换笼周期及刨花垫料使用量的优化研究[J]. 实验动物与比较医学, 2026, 46(2): 251-260. |
| [10] | 李慧. 生物语言学动物模型研究进展[J]. 实验动物与比较医学, 2026, 46(2): 297-305. |
| [11] | 马文静, 刘燕萍, 李一帆, 郭欢, 谢理惠, 杜龙基, 马佳丽, 王红平, 黎家敏. GLP管理体系下实验动物设施消毒剂、饲料和垫料有效期管理实践[J]. 实验动物与比较医学, 2026, (): 1-12. |
| [12] | 王秀然, 李浩, 陈正涛, 于杨, 张素英, 陶如, 王可洲. 医学院校实验动物专业人才培养体系建设的创新与实践[J]. 实验动物与比较医学, 2026, (): 1-10. |
| [13] | 卜纪雯, 华叶, 金仕容, 任宁欣, 李福宁, 杜久林. 斑马鱼品系及斑马鱼房的自动化管理[J]. 实验动物与比较医学, 2026, (): 1-11. |
| [14] | 汤建平, 赵丽亚, 赵莹. 常用近交系大鼠微卫星遗传标记筛选及分析[J]. 实验动物与比较医学, 2026, (): 1-11. |
| [15] | 陈子宜, 孙红燕, 康品方, 武文娟. 肺动脉高压动物模型构建方法及新型技术研究进展[J]. 实验动物与比较医学, 2026, 46(1): 81-93. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||