













实验动物与比较医学 ›› 2026, Vol. 46 ›› Issue (2): 212-221.DOI: 10.12300/j.issn.1674-5817.2025.077
刘赛1, 付彬1, 李思迪1,2, 陈志达1, 张悦1, 郭中坤1, 王永安1( )(
)( ), 王可洲1(
), 王可洲1( )(
)( )
)
收稿日期:2025-05-22
修回日期:2025-09-11
出版日期:2026-04-25
发布日期:2026-04-18
通讯作者:
作者简介:刘 赛(2001—),男,硕士研究生,研究方向:免疫药理学。E-mail:1820739615@qq.com
基金资助:
LIU Sai1, FU Bin1, LI Sidi1,2, CHEN Zhida1, ZHANG Yue1, GUO Zhongkun1, WANG Yongan1( )(
)( ), WANG Kezhou1(
), WANG Kezhou1( )(
)( )
)
Received:2025-05-22
Revised:2025-09-11
Published:2026-04-25
Online:2026-04-18
Contact:
WANG Kezhou (ORCID: 0000-0003-1919-272X), E-mail: wangkezhou_cn@163.com;摘要:
目的 探究肾上腺素受体α2A(adrenoceptor alpha 2A,Adra2a)调节脂多糖(lipopolysaccharide,LPS)诱导脂多糖结合蛋白(lipopolysaccharide-binding protein,LBP)敲除小鼠(Lbp-/- )原代肝细胞炎症的机制。 方法 通过两步灌流法提取C57BL/6J小鼠和Lbp-/- 小鼠(来自C57BL/6J小鼠)原代肝细胞,构建经LPS诱导的原代肝细胞体外炎症模型,同时通过腹腔注射LPS以构建炎症小鼠体内模型。将体外实验分为5组,即Control组、LPS组、BRL+LPS组、OE-NC+LPS组和OE-Adra2a+LPS组。其中,Control组为空白对照;LPS组加入LPS刺激原代肝细胞;BRL+LPS组加入BRL-44408 maleate预处理原代肝细胞后,再加入LPS;OE-NC+LPS组为加入空载病毒转染原代肝细胞后,再加入LPS;OE-Adra2a+LPS组为加入Adra2a过表达慢病毒转染原代肝细胞后,再加入LPS。同样,将体内实验分为5组,即Control'组、LPS'组、BRL+LPS'组、OE-NC+LPS'组、OE-Adra2a+LPS'组。其中,Control'组为空白对照;LPS'组为经腹腔注射LPS;BRL+LPS'组为经腹腔注射BRL-44408 maleate预处理小鼠后,再注射LPS;OE-NC+LPS'组为经腹腔注射空载病毒预处理小鼠后,再注射LPS;OE-Adra2a+LPS'组为经腹腔注射Adra2a过表达慢病毒预处理小鼠后,再注射LPS。通过CCK-8法检测经抑制和过表达Adra2a后的小鼠原代肝细胞存活率,采用实时荧光定量PCR法检测抑制和过表达Adra2a后肿瘤坏死因子-α(tumor necrosis factor-α,TNF-α)、白细胞介素(interleukin,IL)-6和IL-1β这3种炎症因子基因表达量的变化,采用蛋白质印迹法检测经LPS刺激后Adra2a蛋白表达量以及细胞外调节蛋白激酶1/2(extracellular signal-regulated kinase 1/2,ERK1/2)、p38丝裂原激活的蛋白激酶(p38 mitogen-activated protein kinase,p38 MAPK)和c-Jun氨基末端激酶(c-Jun N-terminal kinase,JNK)蛋白磷酸化的变化情况。 结果 体外实验中,在LPS刺激下,与Control组相比,C57BL/6J小鼠原代肝细胞中Adra2a蛋白表达量显著下降(P<0.05),而Lbp-/- 小鼠原代肝细胞中Adra2a蛋白表达量却显著升高(P<0.001);与LPS组相比,BRL+LPS组细胞的存活率显著升高(P<0.01),TNF-α、IL-6和IL-1β基因转录水平显著降低(P<0.01,P<0.001,P<0.001),ERK1/2、p38和JNK蛋白的磷酸化水平均显著降低(P<0.01,P<0.001,P<0.001);与OE-NC+LPS组相比,OE-Adra2a+LPS组细胞存活率显著下降(P<0.001),TNF-α、IL-6和IL-1β基因转录水平显著升高(P<0.001,P<0.01,P<0.001),ERK1/2、p38和JNK蛋白的磷酸化水平均显著升高(P<0.001,P<0.01,P<0.001)。体内实验中,与LPS'组相比,BRL+LPS'组小鼠肝脏组织中ERK1/2、p38和JNK蛋白的磷酸化水平均显著降低(P<0.001,P<0.01,P<0.01);与OE-NC+LPS'组相比,OE-Adra2a+LPS'组小鼠肝脏ERK1/2、p38和JNK蛋白的磷酸化水平均显著升高(P<0.01,P<0.001,P<0.01)。 结论 LPS可引起Lbp-/- 小鼠原代肝细胞Adra2a中蛋白表达量显著升高;Adra2a蛋白可以通过MAPK信号通路调节经LPS诱导的Lbp-/- 小鼠原代肝细胞炎症水平。
中图分类号:
刘赛,付彬,李思迪,等. Adra2a通过MAPK信号通路介导LPS诱导的Lbp-/- 小鼠肝细胞炎性反应[J]. 实验动物与比较医学, 2026, 46(2): 212-221. DOI: 10.12300/j.issn.1674-5817.2025.077.
LIU Sai,FU Bin,LI Sidi,et al. Adra2a Regulates LPS-Induced Inflammation in Hepatocytes of Lbp-/- Mice via the MAPK Signaling Pathway[J]. Laboratory Animal and Comparative Medicine, 2026, 46(2): 212-221. DOI: 10.12300/j.issn.1674-5817.2025.077.
基因 Gene | 正向引物(5’-3’) Forward primer (5’-3’) | 反向引物(5’-3’) Reverse primer (5’-3’) |
|---|---|---|
| Adra2a | GTGACACTGACGCTGGTTTG | CCAGTAACCCATAACCTCGTTG |
| β-actin | GGCTGTATTCCCCTCCATCG | CCAGTTGGTAACAATGCCATGT |
| TNF-α | CCCTCACACTCAGATCATCTTCT | GCTACGACGTGGGCTACAG |
| IL-6 | TAGTCCTTCCTACCCCAATTTCC | TTGGTCCTTAGCCACTCCTTC |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
表1 引物序列
Table 1 Primer sequences
基因 Gene | 正向引物(5’-3’) Forward primer (5’-3’) | 反向引物(5’-3’) Reverse primer (5’-3’) |
|---|---|---|
| Adra2a | GTGACACTGACGCTGGTTTG | CCAGTAACCCATAACCTCGTTG |
| β-actin | GGCTGTATTCCCCTCCATCG | CCAGTTGGTAACAATGCCATGT |
| TNF-α | CCCTCACACTCAGATCATCTTCT | GCTACGACGTGGGCTACAG |
| IL-6 | TAGTCCTTCCTACCCCAATTTCC | TTGGTCCTTAGCCACTCCTTC |
| IL-1β | GCAACTGTTCCTGAACTCAACT | ATCTTTTGGGGTCCGTCAACT |
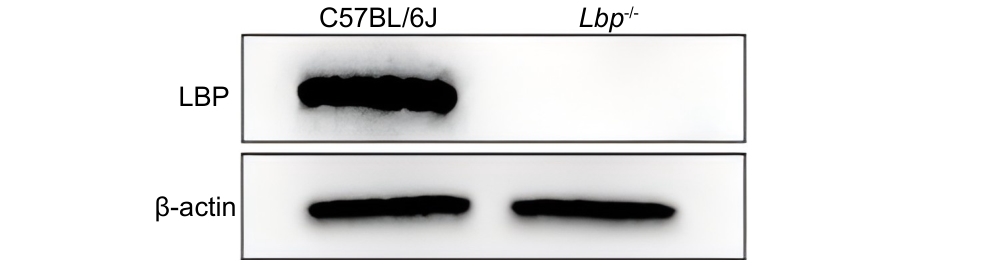
图1 原代肝细胞中LBP蛋白的表达情况
Figure 1 Expression of LBP protein in primary hepatocytes
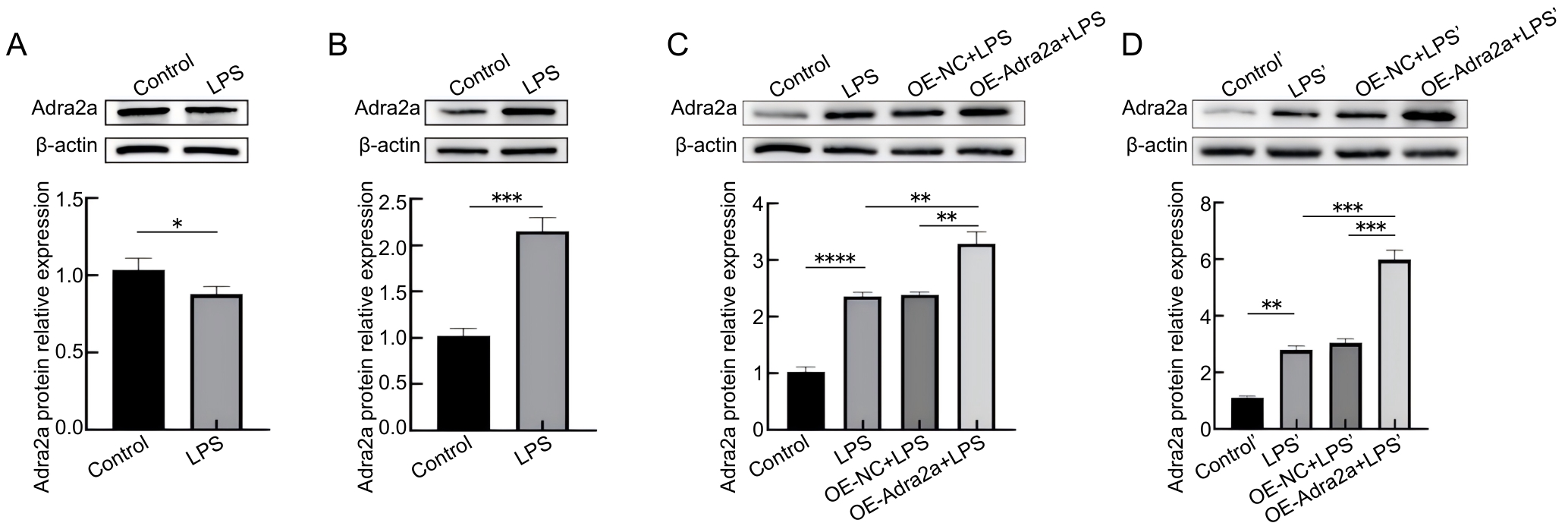
图2 LPS刺激和慢病毒处理后Adra2a蛋白表达的变化
Figure 2 Changes in Adra2a protein expression after LPS and lentivirus treatment
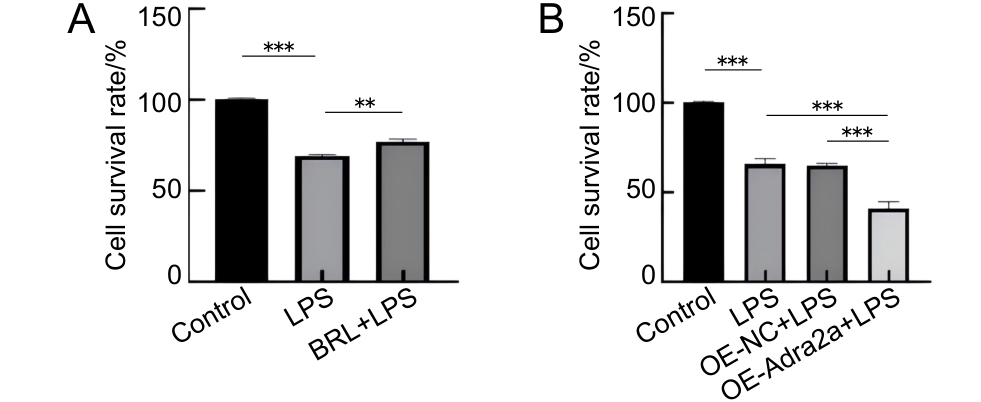
图3 抑制和过表达Adra2a对LPS刺激后Lbp-/- 小鼠原代肝细胞存活率的影响
Figure 3 Effects of Adra2a inhibition and overexpression on the survival rate of primary hepatocytes from Lbp-/- mice following LPS stimulation
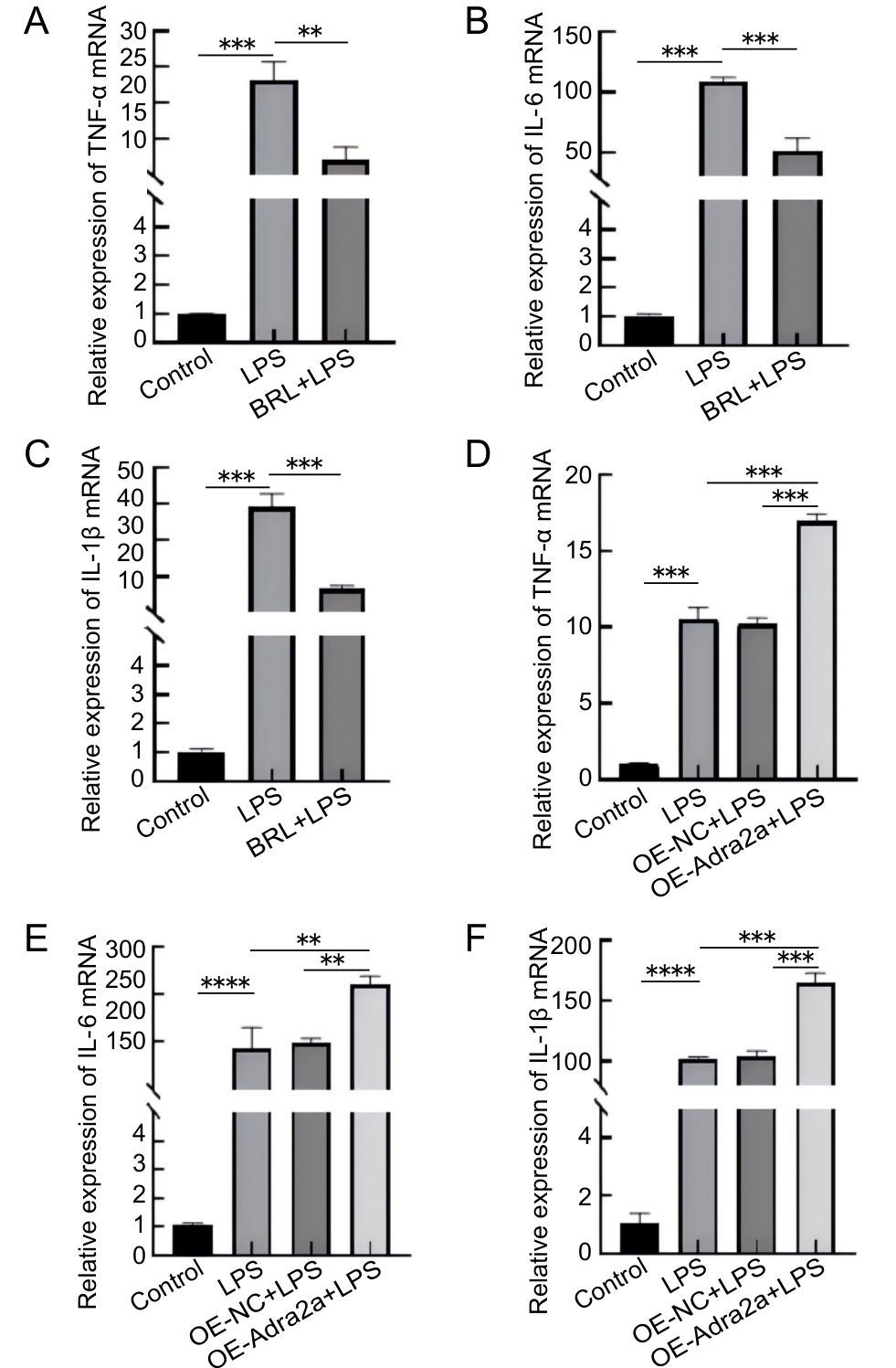
图4 抑制(A~C)和过表达(D~F)Adra2a对LPS刺激后Lbp-/- 小鼠原代肝细胞ΤNF-ɑ、IL-6和IL-1β基因转录的影响
Figure 4 Effects of Adra2a inhibition (A–C) and overexpression (D–F) on the gene transcription of TNF-ɑ, IL-6, and IL-1 β in primary hepatocytes from Lbp-/- mice following LPS stimulation

图5 抑制和过表达Adra2a对Lbp-/- 小鼠原代肝细胞和肝脏组织中ERK1/2、p38、JNK蛋白磷酸化的影响
Figure 5 Effects of Adra2a inhibition and overexpression on ERK1/2, p38, and JNK protein phosphorylation inprimary hepatocytes and liver tissues of Lbp-/- mice
| [1] | DI LORENZO F, DE CASTRO C, SILIPO A, et al. Lipopolysaccharide structures of Gram-negative populations in the gut microbiota and effects on host interactions[J]. FEMS Microbiol Rev, 2019, 43(3):257-272. DOI: 10.1093/femsre/fuz002 . |
| [2] | ULEVITCH R J, TOBIAS P S. Recognition of Gram-negative bacteria and endotoxin by the innate immune system[J]. Curr Opin Immunol, 1999, 11(1):19-22. DOI: 10.1016/S0952-7915(99)80004-1 . |
| [3] | GARCIA-VELLO P, DI LORENZO F, ZUCCHETTA D, et al. Lipopolysaccharide lipid A: a promising molecule for new immunity-based therapies and antibiotics[J]. Pharmacol Ther, 2022, 230:107970. DOI: 10.1016/j.pharmthera.2021.107970 . |
| [4] | SONG Z C, MENG L L, HE Z X, et al. LBP protects hepatocyte mitochondrial function via the PPAR-CYP4A2 signaling pathway in a rat sepsis model[J]. Shock, 2021, 56(6):1066-1079. DOI: 10.1097/SHK.0000000000001808 . |
| [5] | 李思迪, 付彬, 郭中坤, 等. 利用CRISPR/Cas9技术构建脂多糖结合蛋白基因敲除小鼠[J]. 实验动物与比较医学, 2022, 42(4):294-300. DOI: 10.12300/j.issn.1674-5817.2022.002 . |
| LI S D, FU B, GUO Z K, et al. Construction of lipopolysaccaride binding protein knockout mice using CRISPR/Cas9 technology[J]. Lab Anim Comp Med, 2022, 42(4):294-300. DOI: 10.12300/j.issn.1674-5817.2022.002 . | |
| [6] | 米传靓, 付彬, 李思迪, 等. Adra1a调节LPS诱导的Lbp-/-小鼠原代肝细胞炎症反应[J]. 中国比较医学杂志, 2024, 34(5): 84-91. DOI: 10.3969/j.issn.1671-7856.2024.05.009 . |
| MI C L, FU B, LI S D, et al. Adra1a regulates LPS-induced inflammation in primary hepatocytes of Lbp-/- mice[J]. Chin J Comp Med, 2024, 34(5): 84-91. DOI: 10.3969/j.issn.1671-7856.2024.05.009 . | |
| [7] | 米传靓, 付彬, 李思迪, 等. Agtr1a调节LPS诱导的Lbp-/- 小鼠原代肝细胞炎症[J]. 中国实验动物学报, 2023, 31(8): 1021-1027. DOI: 10.3969/j.issn.1005-4847.2023.08.007 . |
| MI C L, FU B, LI S D, et al. Agtr1a regulates LPS-induced inflammation in primary hepatocytes of Lbp-/- mice[J]. Acta Lab Anim Sci Sin, 2023, 31(8): 1021-1027. DOI: 10.3969/j.issn.1005-4847.2023.08.007 . | |
| [8] | ZHOU F, CAI J, CHEN L,et al. ADRA2A contributes to airway inflammation and apoptosis in asthma through the ERK signaling in vitro and in vivo [J]. J Inflamm, 2025, 22(1): 55. DOI: 10.1186/s12950-025-00480-8 . |
| [9] | MIKSA M, DAS P, ZHOU M, et al. Pivotal role of the alpha(2A)-adrenoceptor in producing inflammation and organ injury in a rat model of sepsis[J]. PLoS One, 2009, 4(5):e5504. DOI: 10.1371/journal.pone.0005504 . |
| [10] | LIN Y, ZHU X, YAO W Z, et al. Yohimbine protects against endotoxin-induced acute lung injury by blockade of alpha 2A adrenergic receptor in rats[J]. Chin Med J, 2011, 124(7):1069-1074.DOI: 10.3760/cma.j.issn.0366-6999.2011.07.022 . |
| [11] | 任文洁, 林哲绚. 胰蛋白酶和胶原酶灌注法分离提取小鼠原代肝细胞的比较[J]. 汕头大学医学院学报, 2022, 35(4):204-209. DOI: 10.13401/j.cnki.jsumc.2022.04.003 . |
| REN W J, LIN Z X. Comparison of isolation and extraction of primary mouse hepatocytes by trypsin and collagenase perfusion[J]. J Shantou Univ Med Coll, 2022, 35(4):204-209. DOI: 10.13401/j.cnki.jsumc.2022.04.003 . | |
| [12] | RYU J K, KIM S J, RAH S H, et al. Reconstruction of LPS transfer cascade reveals structural determinants within LBP, CD14, and TLR4-MD2 for efficient LPS recognition and transfer[J]. Immunity, 2017, 46(1):38-50. DOI: 10.1016/j.immuni. 2016.11.007 . |
| [13] | MANO S S, KANEHIRA K, TANIGUCHI A. Comparison of cellular uptake and inflammatory response via toll-like receptor 4 to lipopolysaccharide and titanium dioxide nanoparticles[J]. Int J Mol Sci, 2013, 14(7):13154-13170. DOI: 10.3390/ijms140713154 . |
| [14] | LI T X, BAI J J, DU Y C, et al. Thiamine pretreatment improves endotoxemia-related liver injury and cholestatic complications by regulating galactose metabolism and inhibiting macrophage activation[J]. Int Immunopharmacol, 2022, 108:108892. DOI: 10.1016/j.intimp.2022.108892 . |
| [15] | BRASS D M, SAVOV J D, WHITEHEAD G S, et al. LPS binding protein is important in the airway response to inhaled endotoxin[J]. J Allergy Clin Immunol, 2004, 114(3):586-592. DOI: 10.1016/j.jaci.2004.04.043 . |
| [16] | MINTER R M, BI X M, BEN-JOSEF G, et al. LPS-binding protein mediates LPS-induced liver injury and mortality in the setting of biliary obstruction[J]. Am J Physiol Gastrointest Liver Physiol, 2009, 296(1): G45-G54. DOI: 10.1152/ajpgi.00041.2008 . |
| [17] | LATTIN J, ZIDAR D A, SCHRODER K, et al. G-protein-coupled receptor expression, function, and signaling in macrophages[J]. J Leukoc Biol, 2007, 82(1):16-32. DOI: 10.1189/jlb.0107051 . |
| [18] | SHARMA N, SISTLA R, ANDUGULAPATI S B. Yohimbine ameliorates liver inflammation and fibrosis by regulating oxidative stress and Wnt/β-catenin pathway[J]. Phyto-medicine, 2024, 123:155182. DOI: 10.1016/j.phymed.2023. 155182 . |
| [19] | WANG J H, LIU Y J, GUO Y S, et al. Function and inhibition of P38 MAP kinase signaling: Targeting multiple inflammation diseases[J]. Biochem Pharmacol, 2024, 220:115973. DOI: 10. 1016/j.bcp.2023.115973 . |
| [20] | AHMED T, ZULFIQAR A, ARGUELLES S, et al. Map kinase signaling as therapeutic target for neurodegeneration[J]. Pharmacol Res, 2020, 160:105090. DOI:10.1016/j.phrs. 2020. 105090 . |
| [21] | REZATABAR S, KARIMIAN A, RAMESHKNIA V, et al. RAS/MAPK signaling functions in oxidative stress, DNA damage response and cancer progression[J]. J Cell Physiol, 2019, 234(9):14951-14965. DOI: 10.1002/jcp.28334 . |
| [22] | 刘淑青. 肝细胞SOX9在肝再生中的作用和机制及肝细胞TGF-β信号通路对肝纤维化的影响和机制[D]. 上海: 中国人民解放军海军军医大学, 2023. DOI: 10.26998/d.cnki.gjuyu.2023.000324 . |
| LIU S Q. The role and underlying mechanism of hepatocellular SOX9 in liver regeneration and hepatocellular TGF-β signaling pathway in liver fibrosis[D]. Shanghai: Naval Medical University, 2023. DOI: 10.26998/d.cnki.gjuyu.2023.000324 . |
| [1] | 孙强. 有关仿制药质量监管缺位与FDA新政的思考[J]. 实验动物与比较医学, 2026, 46(2): 306-310. |
| [2] | 赵赫, 张涛, 李丽, 肖宇宙, 安学芳, 张帆. 猪抑制素多克隆抗体对C57BL/6J小鼠超数排卵的增效作用[J]. 实验动物与比较医学, 2026, 46(2): 271-278. |
| [3] | 赵赫, 张涛, 肖宇宙, 李丽, 安学芳, 张帆. 动物生物安全二级实验室猫感染实验的生物风险控制[J]. 实验动物与比较医学, 2026, 46(2): 242-250. |
| [4] | 王晨, 李学波, 贺东华, 常亮堂. 实验狨猴信息管理系统的设计与应用[J]. 实验动物与比较医学, 2026, 46(2): 231-241. |
| [5] | 王莹, 纪文韬, 徐少琼, 陈国元, 冯洁, 吴宝金. 实验动物屏障设施环境微生物污染情况的监测与分析[J]. 实验动物与比较医学, 2026, 46(2): 222-230. |
| [6] | 崔畅婉, 路一平, 于淼, 王爽, 吴思, 孙峥嵘. 溶质载体家族7成员5抑制剂JPH203对单侧输尿管梗阻诱导小鼠肾纤维化的抑制作用[J]. 实验动物与比较医学, 2026, 46(2): 205-211. |
| [7] | 宋静, 杨宗统, 李晓晶, 李自发, 苏凤云, 徐东川, 隋在云. 泻白散对过敏性哮喘大鼠肺、肠组织形态结构及PI3K和Akt表达水平的影响[J]. 实验动物与比较医学, 2026, 46(2): 191-204. |
| [8] | 荣文双, 牛源菲, 刘美婷, 杨梦园, 崔爽, 马丽娜, 富尧, 王连嵋, 曹俊岭. 抗原类型对诱导型干燥综合征小鼠建模的影响[J]. 实验动物与比较医学, 2026, 46(2): 178-190. |
| [9] | 李慧. 生物语言学动物模型研究进展[J]. 实验动物与比较医学, 2026, 46(2): 297-305. |
| [10] | 吴宪文, 刘丽丽, 陈烨, 徐国恒. 繁育体系中育成期小鼠换笼周期及刨花垫料使用量的优化研究[J]. 实验动物与比较医学, 2026, 46(2): 251-260. |
| [11] | 马文静, 刘燕萍, 李一帆, 郭欢, 谢理惠, 杜龙基, 马佳丽, 王红平, 黎家敏. GLP管理体系下实验动物设施消毒剂、饲料和垫料有效期管理实践[J]. 实验动物与比较医学, 2026, (): 1-12. |
| [12] | 王秀然, 李浩, 陈正涛, 于杨, 张素英, 陶如, 王可洲. 医学院校实验动物专业人才培养体系建设的创新与实践[J]. 实验动物与比较医学, 2026, (): 1-10. |
| [13] | 卜纪雯, 华叶, 金仕容, 任宁欣, 李福宁, 杜久林. 斑马鱼品系及斑马鱼房的自动化管理[J]. 实验动物与比较医学, 2026, (): 1-11. |
| [14] | 汤建平, 赵丽亚, 赵莹. 常用近交系大鼠微卫星遗传标记筛选及分析[J]. 实验动物与比较医学, 2026, (): 1-11. |
| [15] | 陈子宜, 孙红燕, 康品方, 武文娟. 肺动脉高压动物模型构建方法及新型技术研究进展[J]. 实验动物与比较医学, 2026, 46(1): 81-93. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||