
Laboratory Animal and Comparative Medicine
• XXXX XXXX •
LI Hongman1( ), CHEN Yutong1, SHI Yingpei1, WANG Yijing1, PAN Yan1, XU Tong3, ZHOU Yi1, DENG Qiyue1, LIU Xue2(
), CHEN Yutong1, SHI Yingpei1, WANG Yijing1, PAN Yan1, XU Tong3, ZHOU Yi1, DENG Qiyue1, LIU Xue2( )(
)( )
)
Online:2026-01-05
Contact:
LIU Xue
LI Hongman,CHEN Yutong,SHI Yingpei,et al. New Perspectives on Mental Health Assessment in Laboratory Animals: Stress Response Monitoring Based on Hair Characteristics[J]. Laboratory Animal and Comparative Medicine. DOI: 10.12300/j.issn.1674-5817.2025.166.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.slarc.org.cn/dwyx/EN/10.12300/j.issn.1674-5817.2025.166
相关领域 Research areas | 影响机制 Mechanisms | 受影响的研究方向 Research directions affected | 偏差示例 Example deviation | 参考文献 References |
|---|---|---|---|---|
Neuroscience | ||||
Immunology | ||||
Pharmacology | ||||
Table 1 Research areas involving the effects of stress on laboratory animals
相关领域 Research areas | 影响机制 Mechanisms | 受影响的研究方向 Research directions affected | 偏差示例 Example deviation | 参考文献 References |
|---|---|---|---|---|
Neuroscience | ||||
Immunology | ||||
Pharmacology | ||||
参数 Parameters | 表现 Display | 相关机制 Related Mechanisms | 参考文献 References | |
|---|---|---|---|---|
| [ | ||||
| [ | ||||
| [ | ||||
| [ | ||||
| [ | ||||
| 毛发强度和弹性降低 | [ | |||
| [ | ||||
| [ | ||||
糖代谢和三羧酸循环 相关代谢物改变 | [ | |||
| [ |
Table 2 Changes on hair characteristics and substance content of laboratory animals caused by stress
参数 Parameters | 表现 Display | 相关机制 Related Mechanisms | 参考文献 References | |
|---|---|---|---|---|
| [ | ||||
| [ | ||||
| [ | ||||
| [ | ||||
| [ | ||||
| 毛发强度和弹性降低 | [ | |||
| [ | ||||
| [ | ||||
糖代谢和三羧酸循环 相关代谢物改变 | [ | |||
| [ |
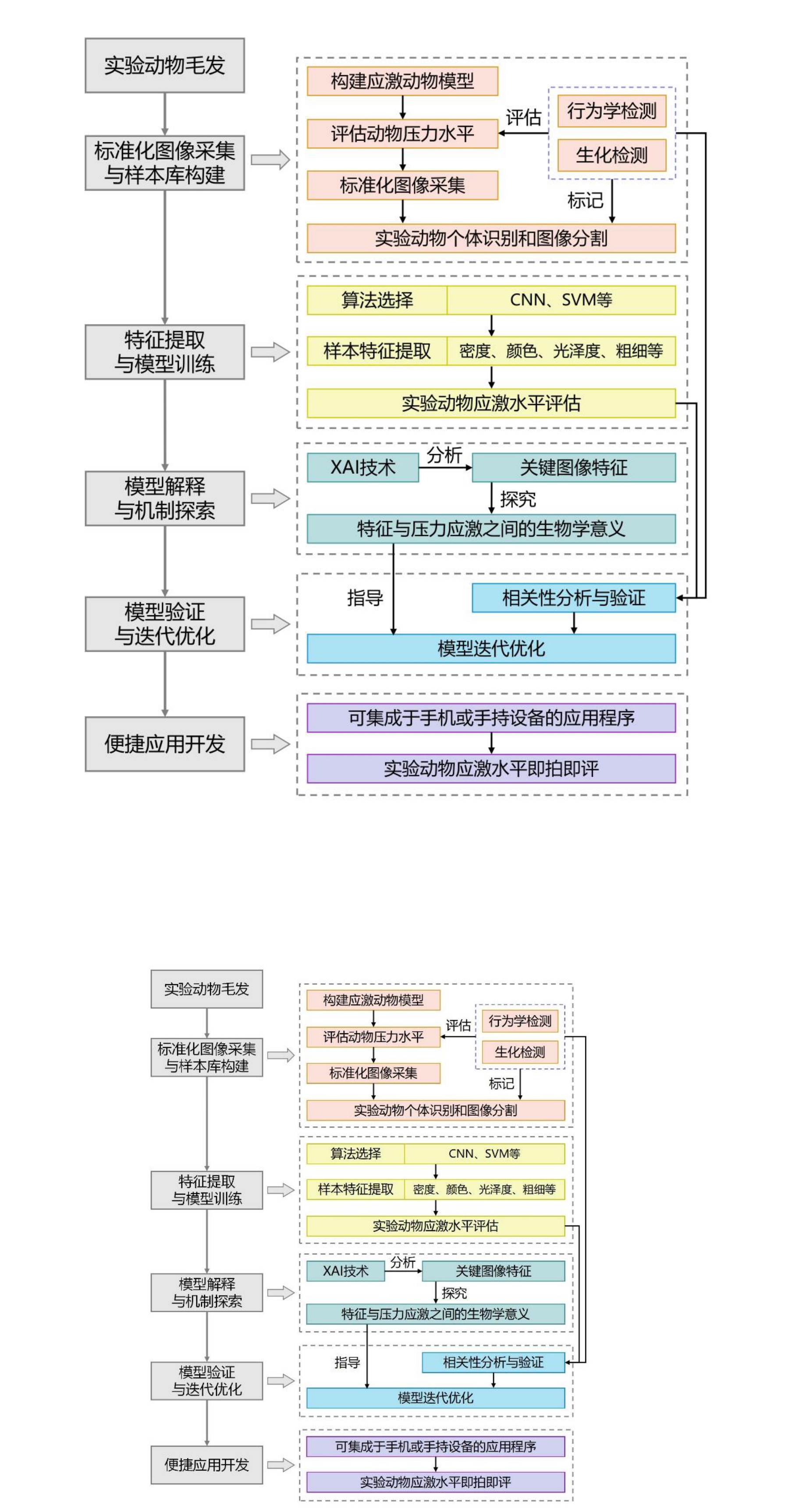
Figure 1 Technical Pathway for Stress Monitoring in Laboratory Animals Based on Hair CharacteristicsNote: CNN:convolutional neural network;SVM:support vector machine;XAI:explainable artificial intelligence.
| [1] | 张静, 刘苗苗, 唐倩倩, 等. 实验动物质量保障体系和服务体系建设[J]. 实验动物科学, 2023, 40(4): 62-68. DOI: 10.3969/j.issn.1006-6179.2023.04.012 . |
| ZHANG J, LIU M M, TANG Q Q, et al. Construction of laboratory animal quality assurance and service system[J]. Lab Anim Sci, 2023, 40(4): 62-68. DOI: 10.3969/j.issn.1006-6179.2023.04.012 . | |
| [2] | The PLOS ONE Staff. Correction: Environmental enrichment effects after early stress on behavior and functional brain networks in adult rats[J]. PLoS One, 2020, 15(4): 1. DOI: 10.1371/journal.pone.0231586 . |
| [3] | TONHAJZEROVA I, MESTANIK M. New perspectives in the model of stress response[J]. Physiol Res, 2017, 66(): S173-S185. DOI: 10.33549/physiolres.933674 . |
| [4] | SHI D D, WANG L P, LI X M, et al. Biological mechanisms of stress-related mental disorders[J]. Sci Sin-Vitae, 2025, 55(7): 1361-1372. DOI: 10.1360/ssv-2024-0346 . |
| [5] | PENG X Q, MAO Y F, TAI Y J, et al. Characterization of anxiety-like behaviors and neural circuitry following chronic moderate noise exposure in mice[J]. Environ Health Perspect, 2023, 131(10): 107004. DOI: 10.1289/EHP12532 . |
| [6] | YANG M, LUO Y, HAO X Z, et al. Effects of high temperature and high humidity stress on the negative feedback regulation of hippocampus on HPA axis in rats[J]. Neuro Endocrinol Lett, 2021, 42(5): 312-320. |
| [7] | HENDERSON L J, SMULDERS T V, ROUGHAN J V. Identifying obstacles preventing the uptake of tunnel handling methods for laboratory mice: an international thematic survey[J]. PLoS One, 2020, 15(4): 1-18. DOI: 10.1371/journal.pone.0231454 . |
| [8] | BRAGA-DIAS A C, CARDENAS OTERO S J, CAVALCANTE OLIVEIRA M K, et al. Social isolation induces sex-differentiated sensory-emotional pain responses and oxytocinergic receptors alterations in mice[J]. Eur J Pain, 2025, 29(8): e70106. DOI: 10.1002/ejp.70106 . |
| [9] | QIN Y H, JIANG X F, LI W, et al. Chronic mild stress leads to aberrant glucose energy metabolism in depressed Macaca fascicularis models[J]. Psychoneuroendocrinology, 2019, 107: 59-69. DOI: 10.1016/j.psyneuen.2019.05.007 . |
| [10] | BALAKIN E, YURKU K, IVANOV M, et al. Regulation of stress-induced immunosuppression in the context of neuroendocrine, cytokine, and cellular processes[J]. Biology, 2025, 14(1): 1-29. DOI: 10.3390/biology14010076 . |
| [11] | WALTHER L M, WIRTZ P H. Physiological reactivity to acute mental stress in essential hypertension—a systematic review[J]. Front Cardiovasc Med, 2023, 10: 1-22. DOI: 10.3389/fcvm.2023.1215710 . |
| [12] | LIGHTMAN S L, BIRNIE M T, CONWAY-CAMPBELL B L. Dynamics of ACTH and cortisol secretion and implications for disease[J]. Endocr Rev, 2020, 41(3): bnaa002. DOI: 10.1210/endrev/bnaa002 . |
| [13] | BEUSCHLEIN F, ELSE T, BANCOS I, et al. European society of endocrinology and endocrine society joint clinical guideline: diagnosis and therapy of glucocorticoid-induced adrenal insufficiency[J]. J Clin Endocrinol Metab, 2024, 109(7): 1657-1683. DOI: 10.1210/clinem/dgae250 . |
| [14] | OBEAGU E I. Stress, neutrophils, and immunity: a dynamic interplay[J]. Ann Med Surg, 2025, 87(6): 3573-3585. DOI: 10.1097/MS9.0000000000003304 . |
| [15] | ODAKA H, ADACHI N, NUMAKAWA T. Impact of glucocorticoid on neurogenesis[J]. Neural Regen Res, 2017, 12(7): 1028-1035. DOI: 10.4103/1673-5374.211174 . |
| [16] | KRASNER H, ONG C V, HEWITT P, et al. From stress to synapse: the neuronal atrophy pathway to mood dysregulation[J]. Int J Mol Sci, 2025, 26(7): 1-37. DOI: 10.3390/ijms26073219 . |
| [17] | KOKKOSIS A G, MADEIRA M M, HAGE Z, et al. Chronic psychosocial stress triggers microglial-/ macrophage-induced inflammatory responses leading to neuronal dysfunction and depressive-related behavior[J]. Glia, 2024, 72(1): 111-132. DOI: 10.1002/glia.24464 . |
| [18] | SHERIDAN J F, FENG N G, BONNEAU R H, et al. Restraint stress differentially affects anti-viral cellular and humoral immune responses in mice[J]. J Neuroimmunol, 1991, 31(3): 245-255. DOI: 10.1016/0165-5728(91)90046-a . |
| [19] | XIA Z C, WEI H Y, DUAN J J, et al. Chronic unpredicted mild stress-induced depression alter saxagliptin pharmacokinetics and CYP450 activity in GK rats[J]. PeerJ, 2016, 4: 1-15. DOI: 10.7717/peerj.1611 . |
| [20] | SAUL A N, OBERYSZYN T M, DAUGHERTY C, et al. Chronic stress and susceptibility to skin cancer[J]. J Natl Cancer Inst, 2005, 97(23): 1760-1767. DOI: 10.1093/jnci/dji401 . |
| [21] | LEI Y R, LIAO F H, TIAN Y C, et al. Investigating the crosstalk between chronic stress and immune cells: implications for enhanced cancer therapy[J]. Front Neurosci, 2023, 17: 1-17. DOI: 10.3389/fnins.2023.1321176 . |
| [22] | YANG H, XIA L, CHEN J, et al. Stress-glucocorticoid-TSC22D3 axis compromises therapy-induced antitumor immunity[J]. Nat Med, 2019, 25(9): 1428-1441. DOI: 10.1038/s41591-019-0566-4 . |
| [23] | DEMORI I, GRASSELLI E. The role of the stress response in metabolic dysfunction-associated fatty liver disease: a psychoneuroendocrineimmunology-based perspective[J]. Nutrients, 2023, 15(3): 1-16. DOI: 10.3390/nu15030795 . |
| [24] | ALONGE M M, GREVILLE L J S, MA X H, et al. Acute restraint stress rapidly impacts reproductive neuroendocrinology and downstream gonad function in big brown bats (Eptesicus fuscus)[J]. J Exp Biol, 2023, 226(19): 1-14. DOI: 10.1242/jeb.245592 . |
| [25] | CUNNINGHAM A M, WALKER D M, RAMAKRISHNAN A, et al. Sperm transcriptional state associated with paternal transmission of stress phenotypes[J]. J Neurosci, 2021, 41(29): 6202-6216. DOI: 10.1523/JNEUROSCI.3192-20.2021 . |
| [26] | SCHUBERT C, LAMBERTZ M, NELESEN R A, et al. Effects of stress on heart rate complexity: a comparison between short-term and chronic stress[J]. Biol Psychol, 2009, 80(3): 325-332. DOI: 10.1016/j.biopsycho.2008.11.005 . |
| [27] | MADISON A A, SHROUT M R, WILSON S J, et al. "I'm Ready This Time": Investigating physiological and emotional habituation to repetitive social stress[J]. Ann Behav Med, 2025, 59(1): kaaf009. DOI: 10.1093/abm/kaaf009 . |
| [28] | YANG X, GONG R, QIN L W, et al. Trafficking of NMDA receptors is essential for hippocampal synaptic plasticity and memory consolidation[J]. Cell Rep, 2022, 40(7): 1-18. DOI: 10.1016/j.celrep.2022.111217 . |
| [29] | GENIS-MENDOZA A D, JUÁREZ-ROJOP I E, ESCOBAR-CHAN Y M, et al. Increased depressive-like, anxiety-like, and perseverative-like behavior in binge eating model in juvenile rats[J]. Nutrients, 2024, 16(9): 1-15. DOI: 10.3390/nu16091275 . |
| [30] | ATAALLAHI M, NEJAD J G, PARK K H. Selection of appropriate biomatrices for studies of chronic stress in animals: a review[J]. J Anim Sci Technol, 2022, 64(4): 621-639. DOI: 10.5187/jast.2022.e38 . |
| [31] | 孙世光, 刘健, 鹿岩, 等. 昆明小鼠强迫游泳实验与悬尾实验抑郁模型相关性[J]. 中国药理学与毒理学杂志, 2014, 28(1): 107-112. DOI: 10.3867/j.issn.1000-3002.2014.01.016 . |
| SUN S G, LIU J, LU Y, et al. Correlation between forced swimming test and tail suspension test in Kunming mice[J]. Chin J Pharmacol Toxicol, 2014, 28(1): 107-112. DOI: 10.3867/j.issn.1000-3002.2014.01.016 . | |
| [32] | ALQURASHI G K, HINDI E A, ZAYED M A, et al. The impact of chronic unpredictable mild stress-induced depression on spatial, recognition and reference memory tasks in mice: behavioral and histological study[J]. Behav Sci, 2022, 12(6): 1-21. DOI: 10.3390/bs12060166 . |
| [33] | BERRIO J P, HESTEHAVE S, KALLIOKOSKI O. Reliability of sucrose preference testing following short or no food and water deprivation-a Systematic Review and Meta-Analysis of rat models of chronic unpredictable stress[J]. Transl Psychiatry, 2024, 14(1): 1-10. DOI: 10.1038/s41398-024-02742-0 . |
| [34] | RINGSEIS R, MARSCHALL M J M, GRUNDMANN S M, et al. Effect of Hermetia illucens fat, compared with that of soybean oil and palm oil, on hepatic lipid metabolism and plasma metabolome in healthy rats[J]. Animals, 2023, 13(21): 1-12. DOI: 10.3390/ani13213356 . |
| [35] | CALABRESE F, GUIDOTTI G, MOLTENI R, et al. Stress-induced changes of hippocampal NMDA receptors: modulation by duloxetine treatment[J]. PLoS One, 2012, 7(5): 1-8. DOI: 10.1371/journal.pone.0037916 . |
| [36] | PANDEY S, HAN W Y, LI J, et al. Reversing anxiety by targeting a stress-responsive signaling pathway[J]. Proc Natl Acad Sci USA, 2024, 121(31): 1-11. DOI: 10.1073/pnas.2400078121 . |
| [37] | EPHRAIM A J. On sudden or rapid whitening of the hair[J]. Arch Dermatol, 1959, 79(2): 228-236. DOI: 10.1001/archderm.1959.01560140090013 . |
| [38] | ZHANG B, MA S, RACHMIN I, et al. Hyperactivation of sympathetic nerves drives depletion of melanocyte stem cells[J]. Nature, 2020, 577(7792): 676-681. DOI: 10.1038/s41586-020-1935-3 . |
| [39] | WANG X H, LIN Y M, YAN L C, et al. Intensive stress impedes hair follicle growth through triggering cell cycle arrest of hair follicle stem cells[J]. FASEB J, 2025, 39(5): e70460. DOI: 10.1096/fj.202403343R . |
| [40] | STALDER T, STEUDTE-SCHMIEDGEN S, ALEXANDER N, et al. Stress-related and basic determinants of hair Cortisol in humans: a meta-analysis[J]. Psychoneuroendocrinology, 2017, 77: 261-274. DOI: 10.1016/j.psyneuen.2016.12.017 . |
| [41] | SHARPLEY C F, MCFARLANE J R, SLOMINSKI A. Stress-linked Cortisol concentrations in hair: what we know and what we need to know[J]. Rev Neurosci, 2011, 23(1): 111-121. DOI: 10.1515/RNS.2011.058 . |
| [42] | QUIST S R, QUIST J. Keep quiet-how stress regulates hair follicle stem cells[J]. Signal Transduct Target Ther, 2021, 6(1): 1-2. DOI: 10.1038/s41392-021-00772-4 . |
| [43] | JI S F, ZHU Z Y, SUN X Y, et al. Functional hair follicle regeneration: an updated review[J]. Signal Transduct Target Ther, 2021, 6(1): 1-11. DOI: 10.1038/s41392-020-00441-y . |
| [44] | HINDS J A, SANCHEZ E R. The role of the hypothalamus–pituitary–adrenal (HPA) axis in test-induced anxiety: assessments, physiological responses, and molecular details[J]. Stresses, 2022, 2(1): 146-155. DOI: 10.3390/stresses2010011 . |
| [45] | OWECKA B, TOMASZEWSKA A, DOBRZENIECKI K, et al. The hormonal background of hair loss in non-scarring alopecias[J]. Biomedicines, 2024, 12(3): 1-14. DOI: 10.3390/biomedicines12030513 . |
| [46] | PANG S L, WU H L, WANG Q, et al. Chronic stress suppresses the expression of cutaneous hypothalamic-pituitary-adrenocortical axis elements and melanogenesis[J]. PLoS One, 2014, 9(5): 1-11. DOI: 10.1371/journal.pone.0098283 . |
| [47] | TRUEB R. Oxidative stress in ageing of hair[J]. Int J Trichol, 2009, 1(1): 6. DOI: 10.4103/0974-7753.51923 . |
| [48] | WANG X W, CAI C Q, LIANG Q C, et al. Integrated transcriptomics and metabolomics analyses of stress-induced murine hair follicle growth inhibition[J]. Front Mol Biosci, 2022, 9: 1-17. DOI: 10.3389/fmolb.2022.781619 . |
| [49] | HADSHIEW I M, FOITZIK K, ARCK P C, et al. Burden of hair loss: stress and the underestimated psychosocial impact of telogen effluvium and androgenetic alopecia[J]. J Invest Dermatol, 2004, 123(3): 455-457. DOI: 10.1111/j.0022-202X.2004.23237.x . |
| [50] | CHOI S, ZHANG B, MA S, et al. Corticosterone inhibits GAS6 to govern hair follicle stem-cell quiescence[J]. Nature, 2021, 592(7854): 428-432. DOI: 10.1038/s41586-021-03417-2 . |
| [51] | ZHANG K X, BAI X F, YUAN Z P, et al. Cellular nanofiber structure with secretory activity-promoting characteristics for multicellular spheroid formation and hair follicle regeneration[J]. ACS Appl Mater Interfaces, 2020, 12(7): 7931-7941. DOI: 10.1021/acsami.9b21125 . |
| [52] | KIM D H, OH S H, CHANG B S. Effects of excessive bleaching on hair: comparative analysis of external morphology and internal microstructure[J]. Appl Microsc, 2024, 54(1): 1-8. DOI: 10.1186/s42649-024-00104-0 . |
| [53] | HARRIS M L, FUFA T D, PALMER J W, et al. A direct link between MITF, innate immunity, and hair graying[J]. PLoS Biol, 2018, 16(5): 1-27. DOI: 10.1371/journal.pbio.2003648 . |
| [54] | LIU N, WANG L H, GUO L L, et al. Chronic restraint stress inhibits hair growth via substance P mediated by reactive oxygen species in mice[J]. PLoS One, 2013, 8(4): 1-11. DOI: 10.1371/journal.pone.0061574 . |
| [55] | AHN D, KIM H, LEE B, et al. Psychological stress-induced pathogenesis of alopecia areata: autoimmune and apoptotic pathways[J]. Int J Mol Sci, 2023, 24(14): 1-15. DOI: 10.3390/ijms241411711 . |
| [56] | CHEN L J, YU Q, GUO F Y, et al. Neurotensin counteracts hair growth inhibition induced by chronic restraint stress[J]. Exp Dermatol, 2024, 33(1): e14990. DOI: 10.1111/exd.14990 . |
| [57] | 吴晓淋, 徐平, 张亚莉, 等. 人体特征的头发代谢组学及蛋白质组学研究进展[J]. 生物工程学报, 2022, 38(10): 3638-3647. DOI: 10.13345/j.cjb.220526 . |
| WU X L, XU P, ZHANG Y L, et al. Distinguishing human characteristics based on hair metabolomics and proteomics: a review[J]. Chin J Biotechnol, 2022, 38(10): 3638-3647. DOI: 10.13345/j.cjb.220526 . | |
| [58] | CHEN Y, GUO J, XING S P, et al. Global-scale metabolomic profiling of human hair for simultaneous monitoring of endogenous metabolome, short- and long-term exposome[J]. Front Chem, 2021, 9: 1-11. DOI: 10.3389/fchem.2021.674265 . |
| [59] | POZEBON D, SCHEFFLER G L, DRESSLER V L. Elemental hair analysis: a review of procedures and applications[J]. Anal Chim Acta, 2017, 992: 1-23. DOI: 10.1016/j.aca.2017.09.017 . |
| [60] | KEMPSON I M, LOMBI E. Hair analysis as a biomonitor for toxicology, disease and health status[J]. Chem Soc Rev, 2011, 40(7): 3915-3940. DOI: 10.1039/c1cs15021a . |
| [61] | SICORELLO M, SPRENGER J C, STÖRKEL L M, et al. Demonstrating the potential of untargeted hair proteomics for personalized biomarkers in stress-associated disorders[J]. Psychoneuroendocrinology, 2026, 184: 107684. DOI: 10.1016/j.psyneuen.2025.107684 . |
| [62] | LAMA N, KASMI R, HAGERTY J R, et al. ChimeraNet: U-Net for hair detection in dermoscopic skin lesion images[J]. J Digit Imag, 2023, 36(2): 526-535. DOI: 10.1007/s10278-022-00740-6 . |
| [63] | URBAN G, FEIL N, CSUKA E, et al. Combining deep learning with optical coherence tomography imaging to determine scalp hair and follicle counts[J]. Lasers Surg Med, 2021, 53(1): 171-178. DOI: 10.1002/lsm.23324 . |
| [64] | ABBAS Q, GARCIA I F, EMRE CELEBI M, et al. A feature-preserving hair removal algorithm for dermoscopy images[J]. Skin Res Technol, 2013, 19(1): e27-e36. DOI: 10.1111/j.1600-0846.2011.00603.x . |
| [65] | GAO M, WANG Y, XU H P, et al. Deep learning-based trichoscopic image analysis and quantitative model for predicting basic and specific classification in male androgenetic alopecia[J]. Acta Derm Venereol, 2022, 102: 1-6. DOI: 10.2340/actadv.v101.564 . |
| [66] | SUN Y K, HUO P J, WANG Y J, et al. Automatic monitoring system for individual dairy cows based on a deep learning framework that provides identification via body parts and estimation of body condition score[J]. J Dairy Sci, 2019, 102(11): 10140-10151. DOI: 10.3168/jds.2018-16164 . |
| [67] | CHU F, ANEX D S, JONES A D, et al. Automated analysis of scanning electron microscopic images for assessment of hair surface damage[J]. R Soc Open Sci, 2020, 7(1): 1-12. DOI: 10.1098/rsos.191438 . |
| [1] | LIU Wentao, LUO Yanhong, LONG Yongxia, LUO Qihui, CHEN Zhengli, LIU Lida. Common Environmental Problems and Testing Experiences in Laboratory Animal Facilities in Sichuan Province [J]. Laboratory Animal and Comparative Medicine, 2025, 45(4): 483-489. |
| [2] | TAN Dengxu, MA Yifan, LIU Ke, ZHANG Yanying, SHI Changhong. Reshaping Intercellular Interactions: Empowering the Exploration of Disease Mechanisms and Therapies Using Organoid Co-Culture Models [J]. Laboratory Animal and Comparative Medicine, 2025, 45(3): 309-317. |
| [3] | SHEN Huangyi, HUANG Yufei, YANG Yunpeng. Research Progress on Characteristics Analysis of Gut Microbiota and Its Sex Differences in Laboratory Animals [J]. Laboratory Animal and Comparative Medicine, 2025, 45(3): 349-359. |
| [4] | XU Shengye, HUANG Junfeng, CHEN Yihang, CHANG Liangtang. Design of a Capture Stress-Free Marmoset Monkey Chair Device for Experiments and Its Preliminary Application [J]. Laboratory Animal and Comparative Medicine, 2025, 45(1): 67-72. |
| [5] | LIU Lida, CHEN Bing, XIE Na, LIU Li, ZHUANG Siqi, ZOU Yixing. Survey Report Analysis on Parasitic and Microbial Quality of Laboratory Animals in Sichuan Province, 2017-2023 [J]. Laboratory Animal and Comparative Medicine, 2024, 44(6): 654-660. |
| [6] | LIU Yishu, CAI Liping. Advances and Challenges of Using Experimental Pigs in Da Vinci Surgical Robot Training [J]. Laboratory Animal and Comparative Medicine, 2024, 44(6): 667-674. |
| [7] | CHEN Bing, ZOU Yixing, WANG Jingdong. Analysis on Current Status and Countermeasures for Laboratory Animal Management in Sichuan Province Based on Administrative Licensing [J]. Laboratory Animal and Comparative Medicine, 2024, 44(5): 560-566. |
| [8] | ZHAO Yong. Three Dimensions of Animal Experiment Ethics: Analysis Based on Value of Life, Animal Welfare, and Risk Prevention [J]. Laboratory Animal and Comparative Medicine, 2024, 44(4): 445-454. |
| [9] | DENG Shaochang, LIN Danrong, LIANG Chujun, LEI Weiqiao, YANG Jinchun, ZHAO Weibo. Analysis of Institutional Characteristics and Implementation of Guangdong Province Laboratory Animals Ordinance in the Past Decade [J]. Laboratory Animal and Comparative Medicine, 2024, 44(4): 455-462. |
| [10] | SUN Qiang. History, Current Status, Challenges and Opportunities of Laboratory Monkey Industry in China [J]. Laboratory Animal and Comparative Medicine, 2024, 44(4): 343-356. |
| [11] | Lingzhi YU, Xiaofeng WEI, Ming LI, Zhihao KONG. Comparison of Methods between Soiled Bedding Sentinels and Exhaust Air Dust PCR for Health Monitoring of Rodent Laboratory Animals [J]. Laboratory Animal and Comparative Medicine, 2024, 44(3): 321-327. |
| [12] | Meitong LIU, Zhang CHEN, Zhaoqiang ZHANG, Di FAN, Zhan HU, Hailing MA. Formulation of Emergency Response Plan for Laboratory Animal Biosafety Emergencies in Hunan Province [J]. Laboratory Animal and Comparative Medicine, 2024, 44(3): 328-334. |
| [13] | Meitong LIU, Zhang CHEN, Zhaoqiang ZHANG, Di FAN, Zhan HU, Hailing MA. Investigation on Current Biosafety Management Status in Laboratory Animal Institutions in Hunan Province [J]. Laboratory Animal and Comparative Medicine, 2024, 44(2): 202-208. |
| [14] | Bing CHEN, Yixing ZOU, Lingyun YAO, Jingdong WANG. Research on Management Policies of Laboratory Animals in Sichuan Province [J]. Laboratory Animal and Comparative Medicine, 2024, 44(2): 209-213. |
| [15] | Jianhua ZHENG, Yunzhi FA, Qiaoyan DONG, Yefeng QIU, Jingqing CHEN. Construction and Evaluation of a Mouse Model with Intestinal Injury by Acute Hypoxic Stress in Plateau [J]. Laboratory Animal and Comparative Medicine, 2024, 44(1): 31-41. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||