
Laboratory Animal and Comparative Medicine ›› 2026, Vol. 46 ›› Issue (1): 32-45.DOI: 10.12300/j.issn.1674-5817.2025.052
• Animal Models of Human Diseases • Previous Articles Next Articles
XU Bo1( ), CHEN Tairen2, FANG Qian1, WU Ji1(
), CHEN Tairen2, FANG Qian1, WU Ji1( )(
)( )
)
Received:2025-03-27
Revised:2025-10-29
Online:2026-02-25
Published:2026-02-14
Contact:
WU Ji
CLC Number:
XU Bo,CHEN Tairen,FANG Qian,et al. Research on Spatiotemporal Gene Expression Profiles and Repair Mechanisms of Spinal Cord Compression and Hemisection Spinal Cord Injury Mouse Models[J]. Laboratory Animal and Comparative Medicine, 2026, 46(1): 32-45. DOI: 10.12300/j.issn.1674-5817.2025.052.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.slarc.org.cn/dwyx/EN/10.12300/j.issn.1674-5817.2025.052
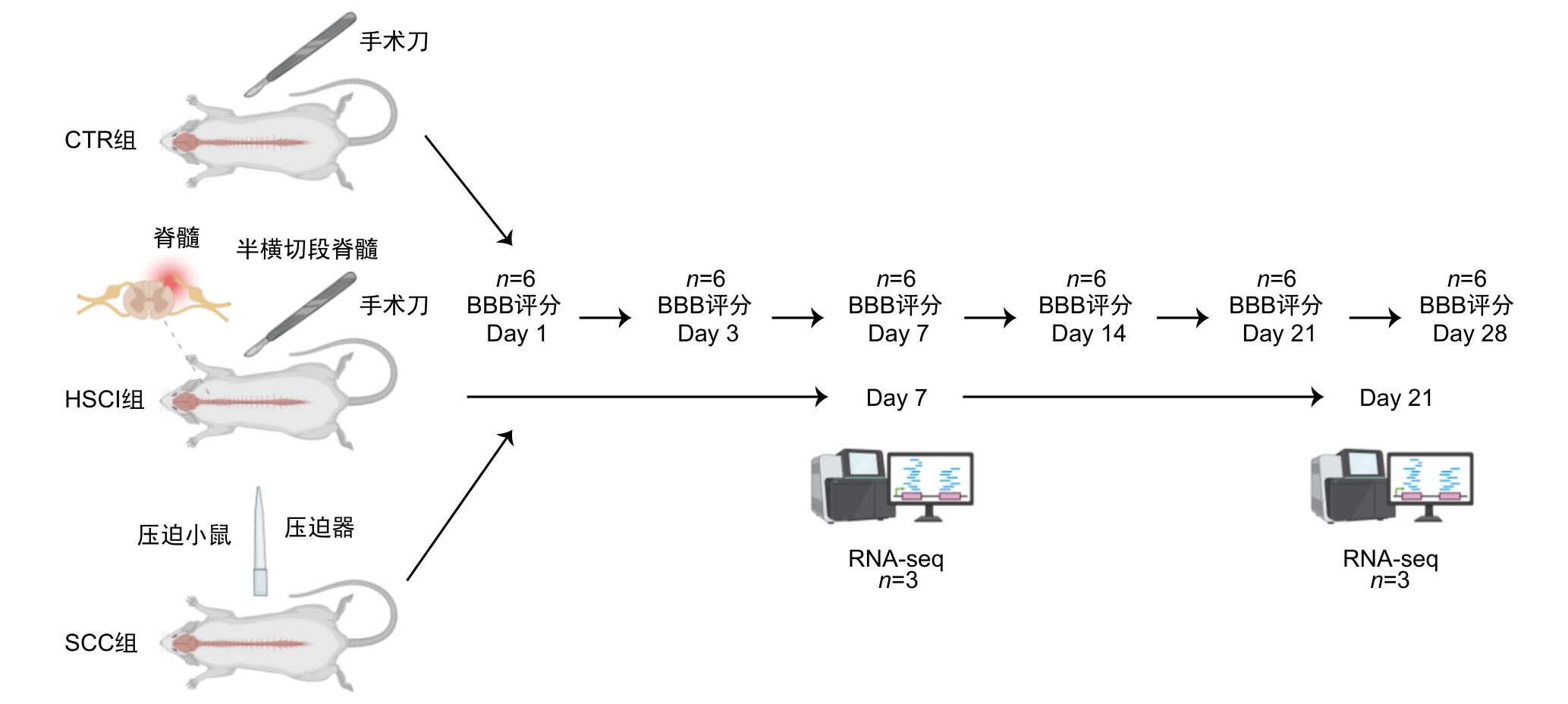
Figure 1 Technical roadmap for model construction and experimental process
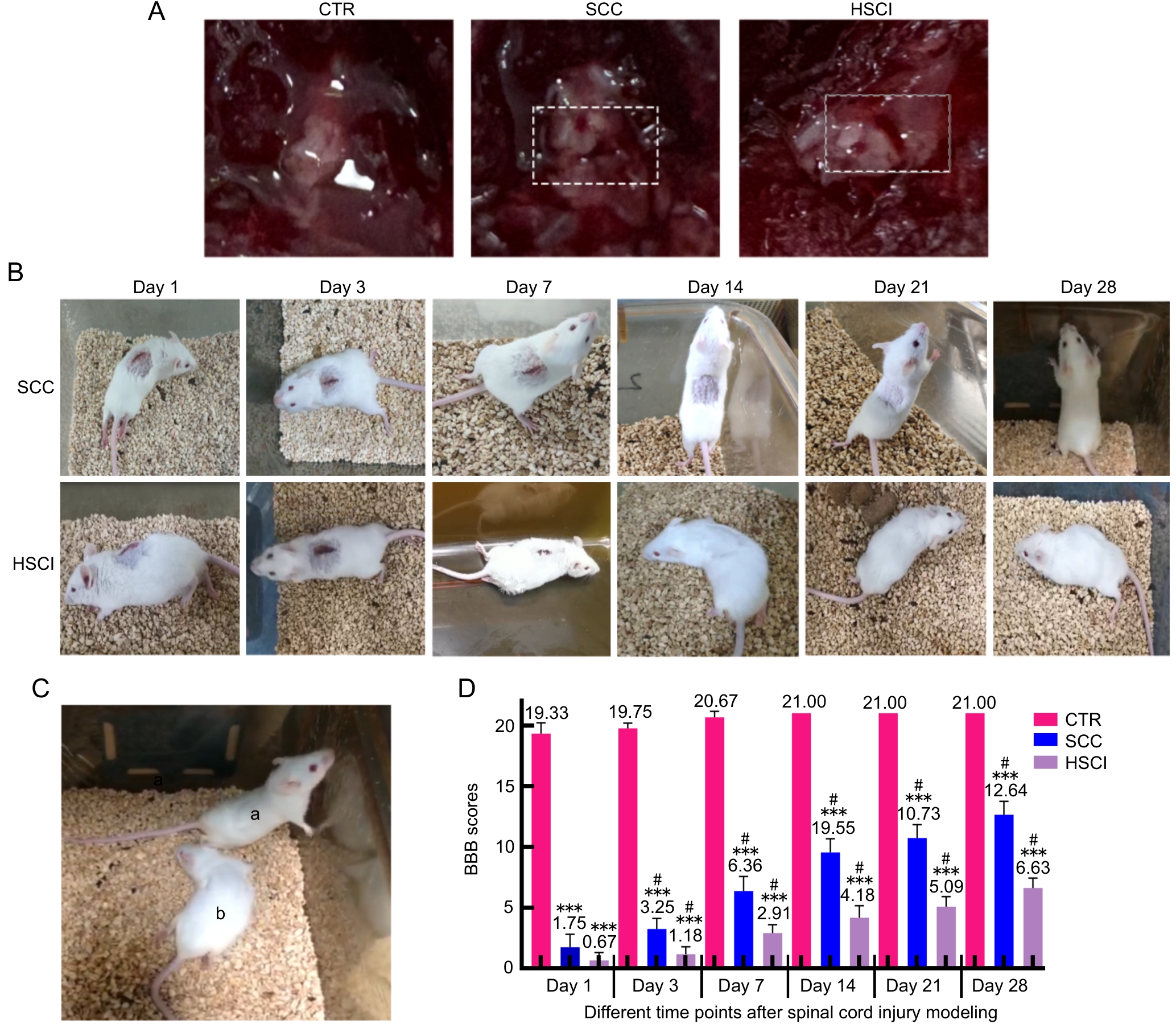
Figure 2 Comparison of macroscopic morphology, recovery process and motor function of mice in sham-operated control group, spinal cord compression and hemisection spinal cord injury group
Note:A, the spinal cord stereoscopy bright-field images of CTR group, SCC group and HSCI group, respectively(the white dashed box indicates the site of spinal cord modeling); B, recovery of mice in SCC and HSCI groups at 1, 3, 7, 14, 21 and 28 days after modeling; C, the comparison chart of the recovery of mice in the SCC group (marked as a) and the HSCI group (marked as b) on the 28th day after surgery; D, BBB scores at different time points after SCI modeling. CTR, sham-operated control group; SCC, spinal cord compression injury group; HSCI, hemisection spinal cord injury group. ***P<0.001, compared with CTR group; #P<0.05, compared with the previous time point within the group.
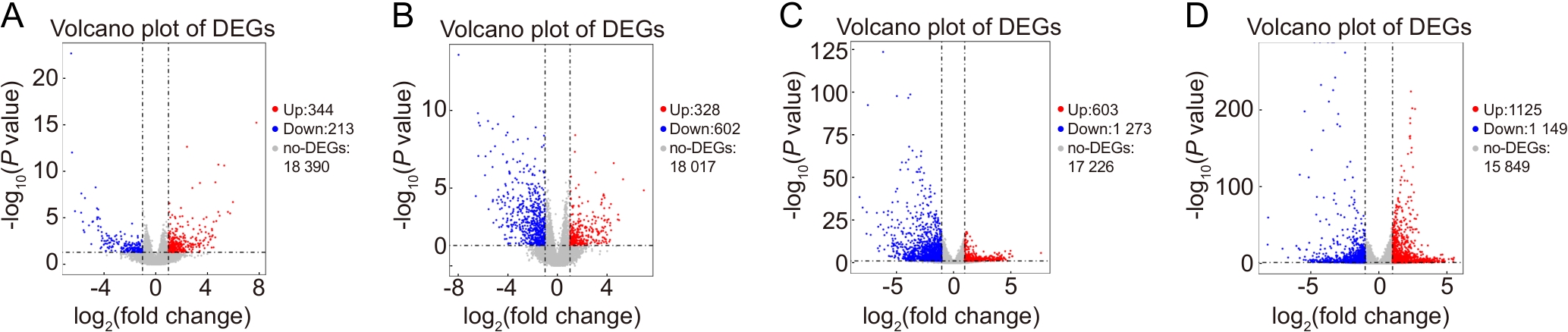
Figure 3 Volcano plots of differentially expressed genes in spinal cord injury models compared with the sham-operated control group at day 7 and day 21 post-injury
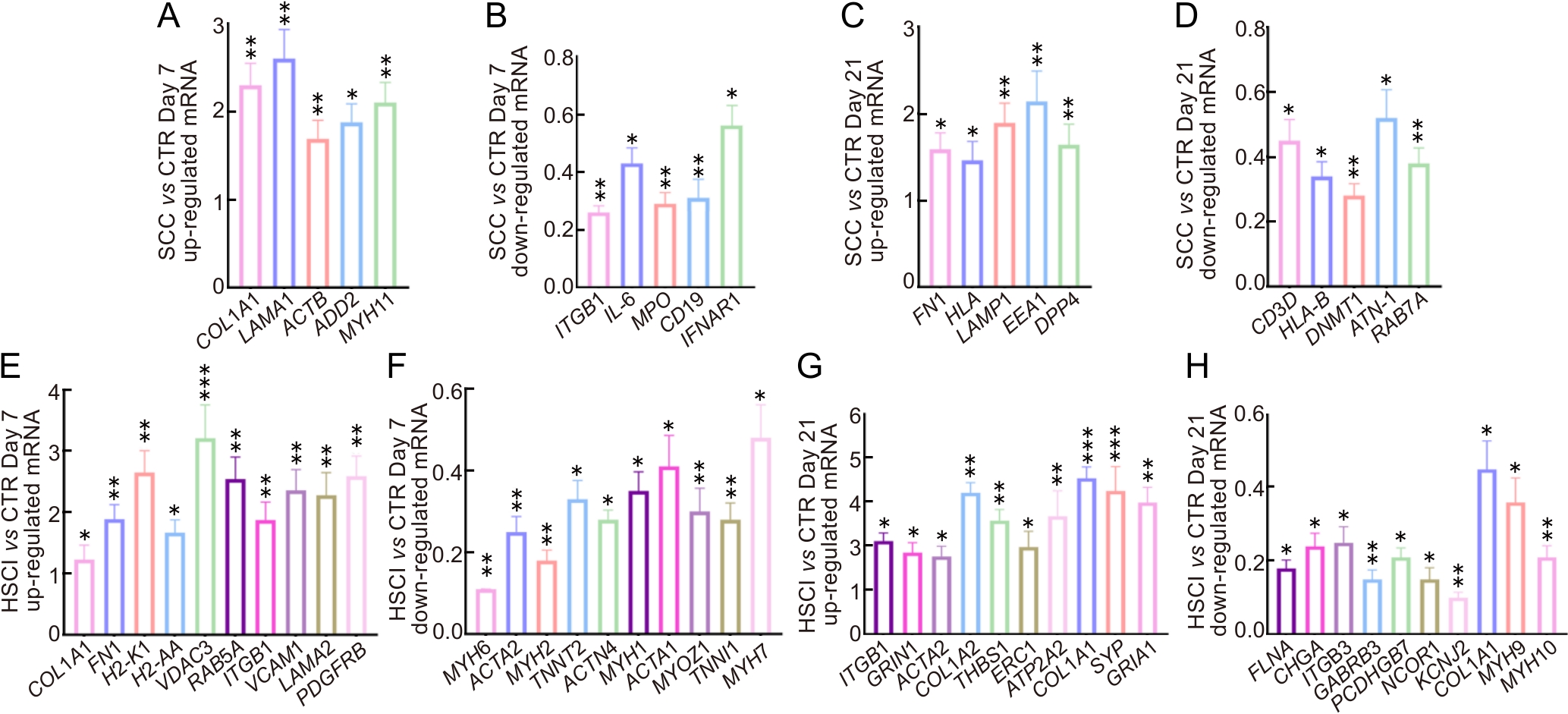
Figure 4 Validation of mRNA expression levels of differentially expressed genes by real time fluorogenic quantitative PCR
Note:A-D, real time fluorogenic quantitative PCR results of the key genes that up-regulated and down-regulated in the SCC group compared with the CTR group at days 7 and 21; E-H, real time fluorogenic quantitative PCR results of the key genes that up-regulated and down-regulated at days 7 and 21 in the HSCI group compared with the CTR group. CTR, sham-operated control group; SCC, spinal cord contusion injury group; HSCI, hemisection spinal cord injury group. All data in the figure are normalized to a baseline of 1 for the CTR group. Real time fluorogenic quantitative PCR experiments were performed using the same batch of mouse tissue samples remaining after RNA-seq(n=3). *P<0.05, **P<0.01, ***P<0.001.

Figure 5 Enrichment analysis of differentially expressed genes in the spinal cord compression group compared to the sham-operated control group at postoperative days 7 and 21
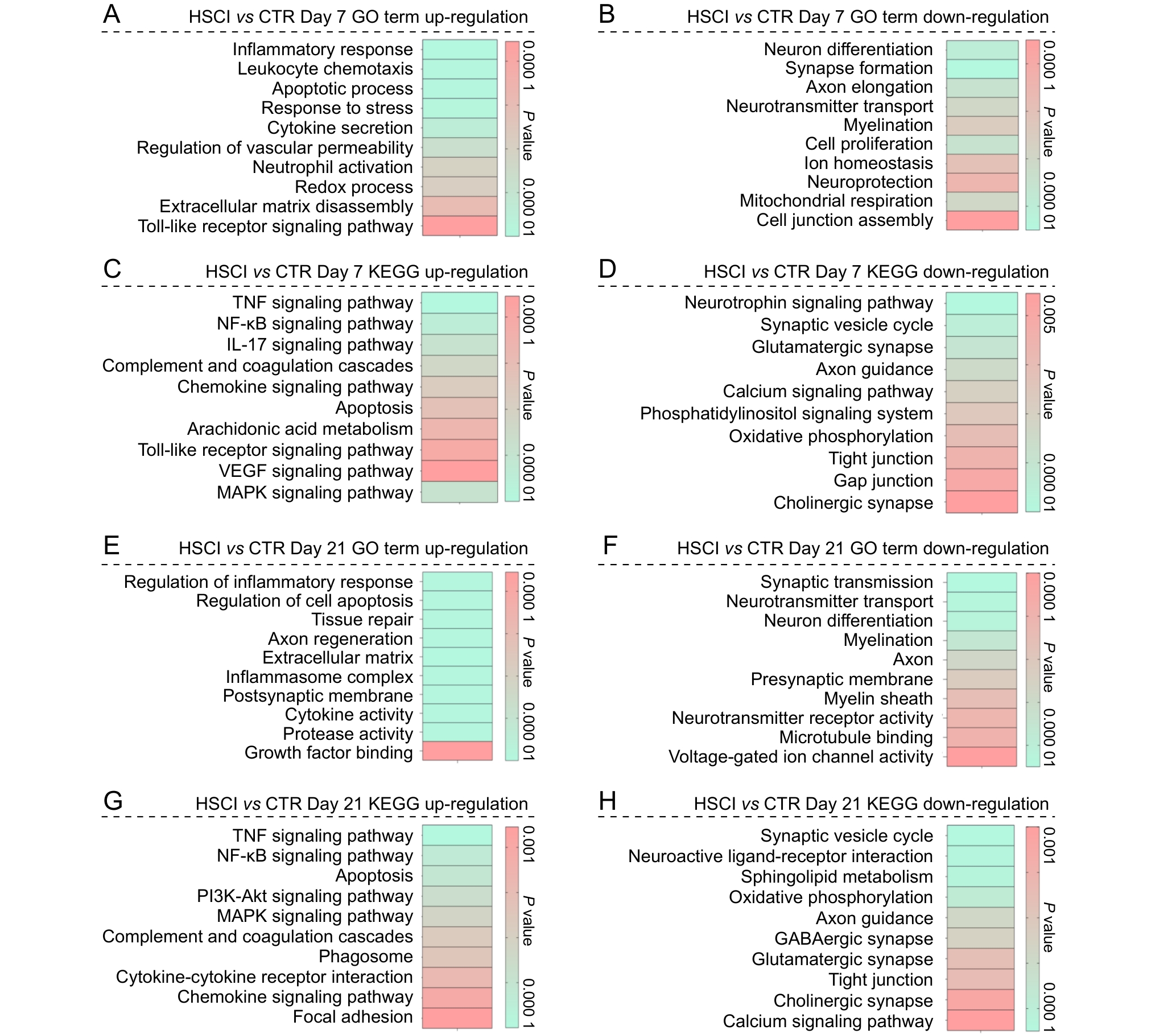
Figure 6 Functional enrichment analysis of differentially expressed genes between the hemisection spinal cord injury group and the sham-operated control group at postoperative days 7 and 21
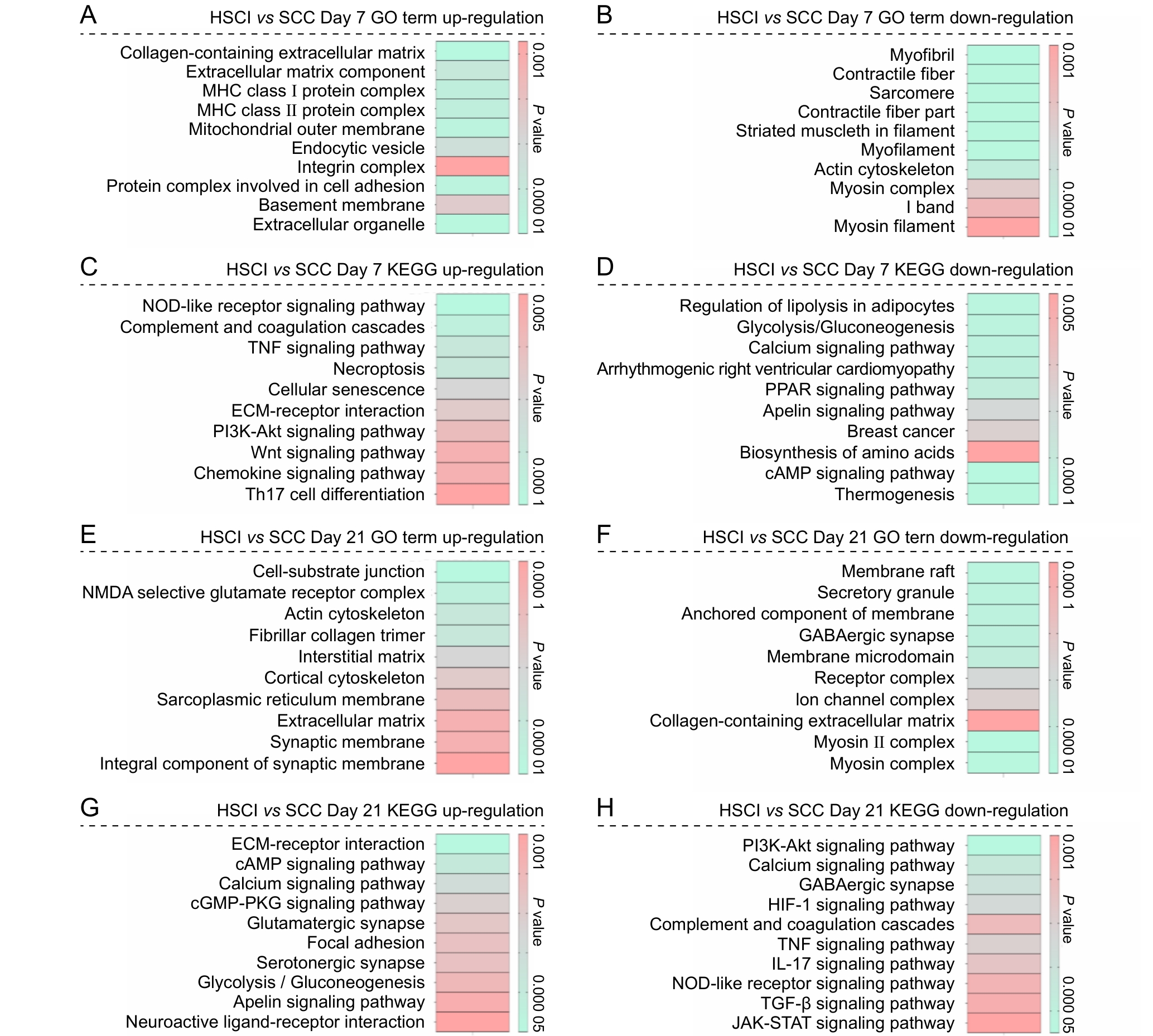
Figure 7 Functional enrichment analysis of differentially expressed genes between the hemisection spinal cord injury group and the spinal cord compression injury group at postoperative days 7 and 21
| [1] | 崔英杰, 孙文卓, 张红梅, 等. 颈椎椎间盘突出致脊髓半横断综合征误诊为脑卒中1例报告[J]. 脊柱外科杂志, 2022, 20(4): 283-285. DOI: 10.3969/j.issn.1672-2957.2022.04.013 . |
| CUI Y J, SUN W Z, ZHANG H M, et al. Brown-Sequard syndrome due to cervical disc herniation misdiagnosed as stroke: a case report[J]. J Spinal Surg, 2022, 20(4): 283-285. DOI: 10.3969/j.issn.1672-2957.2022.04.013 . | |
| [2] | COWAN H, LAKRA C, DESAI M. Autonomic dysreflexia in spinal cord injury[J]. Bmj, 2020: m3596. DOI:10.1136/bmj.m3596 . |
| [3] | FAN B Y, WEI Z J, YAO X, et al. Microenvironment imbalance of spinal cord injury[J]. Cell Transplant, 2018, 27(6):853-866. DOI:10.1177/0963689718755778 . |
| [4] | BASSO D M, FISHER L C, ANDERSON A J, et al. Basso Mouse Scale for locomotion detects differences in recovery after spinal cord injury in five common mouse strains[J]. J Neurotrauma, 2006, 23(5):635-659. DOI:10.1089/neu.2006. 23.635 . |
| [5] | ASHAMMAKHI N, KIM H J, EHSANIPOUR A, et al. Regenerative therapies for spinal cord injury[J]. Tissue Eng Part B Rev, 2019, 25(6):471-491. DOI:10.1089/ten.teb.2019.0182 . |
| [6] | DAVID G, PFYFFER D, VALLOTTON K, et al. Longitudinal changes of spinal cord grey and white matter following spinal cord injury[J]. J Neurol Neurosurg Psychiatry, 2021, 92(11):1222-1230. DOI:10.1136/jnnp-2021-326337 . |
| [7] | ZÜCHNER M, GLOVER J C, BOULLAND J L. A neonatal mouse spinal cord compression injury model[J]. JoVE, 2016(109): 1-10. DOI:10.3791/53498 |
| [8] | ZHU S P, YING Y B, WU Q J, et al. Alginate self-adhesive hydrogel combined with dental pulp stem cells and FGF21 repairs hemisection spinal cord injury via apoptosis and autophagy mechanisms[J]. Chem Eng J, 2021, 426:130827. DOI:10.1016/j.cej.2021.130827 . |
| [9] | EVANS R M, DAVIES M. ScoreCentre: a computer program to assist with collection and calculation of BBB locomotor scale data[J]. J Neurosci Methods, 2010, 194(1):102-107. DOI:10.1016/j.jneumeth.2010.09.018 . |
| [10] | OTANI M, KUSHIDA Y, KURODA Y, et al. New rat model of spinal cord infarction with long-lasting functional disabilities generated by intraspinal injection of endothelin-1[J]. Stroke Vasc Neurol, 2025, 10(2): 1-10. DOI:10.1136/svn-2023-002962 . |
| [11] | WILKINS N, SKINNER N P, MOTOVYLYAK A, et al. Evolution of magnetic resonance imaging as predictors and correlates of functional outcome after spinal cord contusion injury in the rat[J]. J Neurotrauma, 2020, 37(6):889-898. DOI:10.1089/neu.2019.6731 . |
| [12] | NING X J, LU X H, LUO J C, et al. Molecular mechanism of microRNA-21 promoting Schwann cell proliferation and axon regeneration during injured nerve repair[J]. RNA Biol, 2020, 17(10):1508-1519. DOI:10.1080/15476286.2020.1777767 . |
| [13] | MAGGIORE J C, BURRELL J C, BROWNE K D, et al. Tissue engineered axon-based "living scaffolds" promote survival of spinal cord motor neurons following peripheral nerve repair[J]. J Tissue Eng Regen Med, 2020, 14(12):1892-1907. DOI:10.1002/term.3145 . |
| [14] | HU X, XU W, REN Y L, et al. Spinal cord injury: molecular mechanisms and therapeutic interventions[J]. Signal Transduct Target Ther, 2023, 8:1-28. DOI:10.1038/s41392-023-01477-6 . |
| [15] | FAN L, LIU C, CHEN X X, et al. Exosomes-loaded electroconductive hydrogel synergistically promotes tissue repair after spinal cord injury via immunoregulation and enhancement of myelinated axon growth[J]. Adv Sci, 2022, 9(13):1-22. DOI:10.1002/advs.202105586 . |
| [16] | QIAN D F, XU J Q, ZHANG X L, et al. Microenvironment self-adaptive nanomedicine promotes spinal cord repair by suppressing inflammation cascade and neural apoptosis[J]. Adv Mater, 2024, 36(50): e2307624. DOI:10.1002/adma. 202307624 . |
| [17] | MA J J, LI J Y, WANG X R, et al. GDNF-loaded polydopamine nanoparticles-based anisotropic scaffolds promote spinal cord repair by modulating inhibitory microenvironment[J]. Adv Healthc Mater, 2023, 12(8): e2202377. DOI:10.1002/adhm.202202377 . |
| [18] | GAO L S, WANG C, QIN B, et al. 6-phosphofructo-2-kinase/fructose-2, 6-bisphosphatase Suppresses Neuronal Apoptosis by Increasing Glycolysis and "cyclin-dependent kinase 1-Mediated Phosphorylation of p27 After Traumatic Spinal Cord Injury in Rats[J]. Cell Transplant, 2020, 29:1-14. DOI:10.1177/0963689720950226 . |
| [19] | MATA-BERMUDEZ A, RÍOS C, BURELO M, et al. Amantadine prevented hypersensitivity and decreased oxidative stress by NMDA receptor antagonism after spinal cord injury in rats[J]. Eur J Pain, 2021, 25(8):1839-1851. DOI:10.1002/ejp.1795 . |
| [20] | ZHOU W J, ZHAO T T, DU J Y, et al. TIGAR promotes neural stem cell differentiation through acetyl-CoA-mediated histone acetylation[J]. Cell Death Dis, 2019, 10:1-13. DOI:10.1038/s41419-019-1434-3 . |
| [21] | ZHAO J, CHEN K Z, WANG T, et al. Vimentin inhibits neuronal apoptosis after spinal cord injury by enhancing autophagy[J]. CNS Neurosci Ther, 2025, 31(1): 1-14. DOI:10.1111/cns.70200 . |
| [22] | YAO R B, REN L R, WANG S Y, et al. Euxanthone inhibits traumatic spinal cord injury via anti-oxidative stress and suppression of p38 and PI3K/Akt signaling pathway in a rat model[J]. Transl Neurosci, 2021, 12(1):114-126. DOI:10.1515/tnsci-2021-0012 . |
| [23] | CAO J F, HU X, XIONG L, et al. Interference of interleukin-1β mediated by lentivirus promotes functional recovery of spinal cord contusion injury in rats via the PI3K/AKT1 signaling pathway[J]. Mediat Inflamm, 2022, 2022(1):1-17. DOI:10.1155/2022/6285099 . |
| [24] | WANG Y M, ZHAO Z, REGE S V, et al. 3K3A-activated protein C stimulates postischemic neuronal repair by human neural stem cells in mice[J]. Nat Med, 2016, 22(9):1050-1055. DOI:10.1038/nm.4154 . |
| [1] | HE Jiaqi, ZHOU Yuanyuan, NIE Yongqiang, WANG Zhaoxia, XU Wangjie. Establishment and Multidimensional Pathological Evaluations of a Cigarette Smoke Exposure-Induced Chronic Obstructive Pulmonary Disease Mouse Model [J]. Laboratory Animal and Comparative Medicine, 2026, 46(1): 11-19. |
| [2] | GONG Leilei, WANG Xiaoxia, FENG Xuewei, LI Xinlei, ZHAO Han, ZHANG Xueyan, FENG Xin. A Mouse Model and Mechanism Study of Premature Ovarian Insufficiency Induced by Different Concentrations of Cyclophosphamide [J]. Laboratory Animal and Comparative Medicine, 2025, 45(4): 403-410. |
| [3] | JIANG Juan, SONG Ning, LIAN Wenbo, SHAO Congcong, GU Wenwen, SHI Yan. Comparison of Histopathological and Molecular Pathological Phenotypes in Mouse Models of Intrauterine Adhesions Induced by Two Concentrations of Ethanol Perfusion [J]. Laboratory Animal and Comparative Medicine, 2025, 45(4): 393-402. |
| [4] | LUO Lianlian, YUAN Yanchun, WANG Junling, SHI Guangsen. Advances in Mouse Models of Amyotrophic Lateral Sclerosis [J]. Laboratory Animal and Comparative Medicine, 2025, 45(3): 290-299. |
| [5] | QI Longju, CHEN Shiyuan, LIAO Zehua, SHI Yuanhu, SUN Yuyu, WANG Qinghua. Transcriptomic Analysis of Menstrual Blood-Derived Stem Cells Transplantation Combined with Exercise Training in Promoting Spinal Cord Injury Recovery in Rats [J]. Laboratory Animal and Comparative Medicine, 2024, 44(5): 531-542. |
| [6] | Yuan ZHANG, Han LI, Chengfang ZHANG. Whole-brain Transcriptomic Analysis of Weight Gain Mice induced by Olanzapine [J]. Laboratory Animal and Comparative Medicine, 2023, 43(3): 262-270. |
| [7] | Jingwei MA, Gen LI, Yang YANG, Caixia ZANG, Xiuqi BAO, Dan ZHANG. Comparative Study on Different Recovery Periods of the Spermatogenic Dysfunction Mouse Model Induced by Cyclophosphamide [J]. Laboratory Animal and Comparative Medicine, 2023, 43(2): 112-123. |
| [8] | Shiyan YU. Advances in the Application of Mouse Models to Study Digestive Mucosal Immunity and Infectious Diseases [J]. Laboratory Animal and Comparative Medicine, 2022, 42(1): 3-10. |
| [9] | YAN Yitong, BAI Fan, JING Yingli, WANG Limiao, LI Zihan, YU Yan. A Comparative Study on Spontaneous Homecage Behaviors of Spinal Cord Injury Mice [J]. Laboratory Animal and Comparative Medicine, 2021, 41(4): 290-298. |
| [10] | YAO Ding, ZHOU Jing, YAN Guofeng, WANG Huiyang, WANG Yadi, MA Zhengwen. Establishment of Salt-sensitive Hypertension Model in C57BL/6J Mice [J]. Laboratory Animal and Comparative Medicine, 2020, 40(4): 314-. |
| [11] | ZHENG Jie, ZHANG Guozhong, HU Yanmei, WU Xueyi, YANG Zongfu, ZI Xiangdong. Single Nucleotide Polymorphism and Alternative Splicing Analysis between Fresh and Vitrified-thawed Crossbred Blastocysts of Yak via RNA-Seq [J]. Laboratory Animal and Comparative Medicine, 2020, 40(4): 279-. |
| [12] | CHAI Wenjun, SUN Lei, LIU Xiaoli, PAN Hongyu, GUO Tianan, XU Ye, YAN Mingxia. Establishment of Bone Metastasis Mouse Models through Injecting Human Lung Cancer Cells into Left Ventricle#br# under Ultrasound Guidance#br# [J]. Laboratory Animal and Comparative Medicine, 2020, 40(3): 183-. |
| [13] | JIANG Hongli, MA Hongye, XUE Jinhong, SUN Lingshuang, CHEN Lei. The Role of Villin-1 in Model of Habu Nephritis Mice with Unilateral Nephrectomy [J]. Laboratory Animal and Comparative Medicine, 2020, 40(1): 1-8. |
| [14] | HE Yi-min, GU Ming-min. Preliminary Phenotypic Analysis of Myh13 Knockout Mouse [J]. Laboratory Animal and Comparative Medicine, 2019, 39(3): 193-200. |
| [15] | SHEN Yan, XU Wang-yang, ZHU Hou-bao. Research Progress on Pathogenesis of Hereditary Diseases Caused by Mutations of Oxidoreductase DHTKD1 and Related Mouse Models [J]. Laboratory Animal and Comparative Medicine, 2018, 38(6): 468-472. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||