
Laboratory Animal and Comparative Medicine ›› 2026, Vol. 46 ›› Issue (2): 153-177.DOI: 10.12300/j.issn.1674-5817.2025.073
• Animal Models of Human Diseases • Next Articles
 ), CHEN Lin(
), CHEN Lin( ), CHEN Zhiguo(
), CHEN Zhiguo( ), LU Ming(
), LU Ming( ), LI Yingjun(
), LI Yingjun( )
)
Received:2025-05-15
Revised:2025-08-15
Online:2026-04-25
Published:2026-04-18
Contact:
HAN Fabin, CHEN Lin, CHEN Zhiguo, LU Ming, LI Yingjun
CLC Number:
HAN Fabin,CHEN Lin,CHEN Zhiguo,et al. Guidelines for Selecting Preclinical Animal Models for Drugs and Stem Cell Therapies for Parkinson Disease (2026 Edition)[J]. Laboratory Animal and Comparative Medicine, 2026, 46(2): 153-177. DOI: 10.12300/j.issn.1674-5817.2025.073.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.slarc.org.cn/dwyx/EN/10.12300/j.issn.1674-5817.2025.073
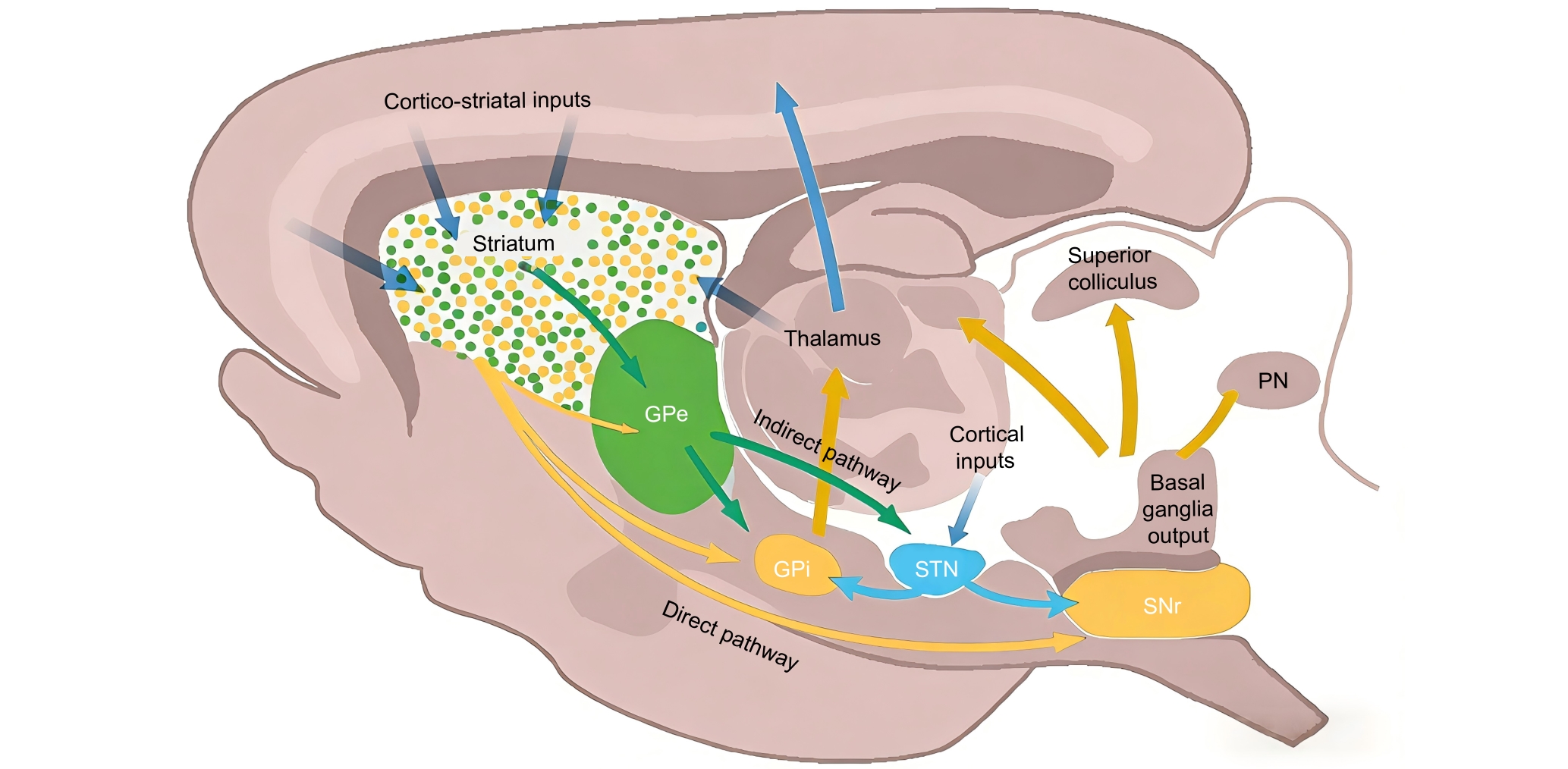
Figure 1 The projection pathway of striatal dopaminergic neurons in the direct and indirect pathways of the basal ganglia
分类 Classification | 症状 Symptom | 出现阶段 Stages of emergence | 临床表现 Clinical manifestation |
|---|---|---|---|
运动症状 Motor symptoms | 运动迟缓 | PD早期 | 自发运动普遍缓慢,手臂摆动、面部表情和手势减少,床上翻身困难,声音低沉;自主重复运动的速度和幅度逐渐降低,包括手指敲击、握力、旋前-旋后运动、脚趾敲击和足跟踩踏 |
| 肌强直 | PD早期 | 肌肉僵硬 | |
| 震颤 | PD早期 | 静止性震颤常发生于四肢、嘴唇和下颌,很少发生在头部。在以目标为导向的随意运动中,震颤幅度减少或消失;检查手部震颤时,患者处于坐位,双手放松,双臂支撑在大腿部 | |
| 步态改变 | PD早期 | 手臂摆动幅度减小,拖着一条腿,走路时姿势微弯 | |
| 姿势改变 | PD后期 | 站立时,躯干向前弯曲,双臂外展,肘部弯曲 | |
| 冻结步态 | PD后期 | 在行走开始、行走期间、转弯或接近狭窄空间时,突然或短暂地无法进行有效向前迈步 | |
| 平衡运动 | PD后期 | 站立和行走时身体不稳,容易跌倒 | |
| 构音障碍和吞咽困难 | PD后期 | 语言障碍,进食困难 | |
非运动症状 Non-motor symptoms | 嗅觉减退 | PD早期 | 多达70%患者表现为嗅觉丧失 |
| 睡眠障碍 | PD早期 | 快速眼动睡眠行为障碍,失眠,周期性肢体运动,静坐不能,白天嗜睡等 | |
| 精神科特征 | PD早期 | 明显的冷漠、焦虑、抑郁 | |
| 自主神经功能障碍 | PD早期 | 便秘,胃排空延迟,尿急或尿失禁,勃起功能障碍,直立性低血压,怕热 | |
| 轻度认知障碍 | PD早期 | 注意力和认知能力轻度下降 | |
| 疼痛和躯体感觉障碍 | PD早期 | 疼痛,感觉异常和烧灼感 | |
| 痴呆 | PD后期 | 约30%患者会出现痴呆的症状,该症状的发病率随着PD病程的发展而增加 |
Table 1 Motor and non-motor symptoms in early and late stages of Parkinson disease(PD)
分类 Classification | 症状 Symptom | 出现阶段 Stages of emergence | 临床表现 Clinical manifestation |
|---|---|---|---|
运动症状 Motor symptoms | 运动迟缓 | PD早期 | 自发运动普遍缓慢,手臂摆动、面部表情和手势减少,床上翻身困难,声音低沉;自主重复运动的速度和幅度逐渐降低,包括手指敲击、握力、旋前-旋后运动、脚趾敲击和足跟踩踏 |
| 肌强直 | PD早期 | 肌肉僵硬 | |
| 震颤 | PD早期 | 静止性震颤常发生于四肢、嘴唇和下颌,很少发生在头部。在以目标为导向的随意运动中,震颤幅度减少或消失;检查手部震颤时,患者处于坐位,双手放松,双臂支撑在大腿部 | |
| 步态改变 | PD早期 | 手臂摆动幅度减小,拖着一条腿,走路时姿势微弯 | |
| 姿势改变 | PD后期 | 站立时,躯干向前弯曲,双臂外展,肘部弯曲 | |
| 冻结步态 | PD后期 | 在行走开始、行走期间、转弯或接近狭窄空间时,突然或短暂地无法进行有效向前迈步 | |
| 平衡运动 | PD后期 | 站立和行走时身体不稳,容易跌倒 | |
| 构音障碍和吞咽困难 | PD后期 | 语言障碍,进食困难 | |
非运动症状 Non-motor symptoms | 嗅觉减退 | PD早期 | 多达70%患者表现为嗅觉丧失 |
| 睡眠障碍 | PD早期 | 快速眼动睡眠行为障碍,失眠,周期性肢体运动,静坐不能,白天嗜睡等 | |
| 精神科特征 | PD早期 | 明显的冷漠、焦虑、抑郁 | |
| 自主神经功能障碍 | PD早期 | 便秘,胃排空延迟,尿急或尿失禁,勃起功能障碍,直立性低血压,怕热 | |
| 轻度认知障碍 | PD早期 | 注意力和认知能力轻度下降 | |
| 疼痛和躯体感觉障碍 | PD早期 | 疼痛,感觉异常和烧灼感 | |
| 痴呆 | PD后期 | 约30%患者会出现痴呆的症状,该症状的发病率随着PD病程的发展而增加 |
α突触核蛋白基因 α-synuclein | 启动子 Promoter | 品种品系 Breed and strain | 多巴胺能神经元的损失率 Loss rate of dopaminergic neurons | α突触核蛋白的病理表型 Pathological phenotype of α-synuclein | 运动表型 Motor phenotype | 非运动表型 Non-motor phenotype |
|---|---|---|---|---|---|---|
| Gene fragments(1~130)[ | Thy-1 | C57BL/6J小鼠 | 约50% | 异常聚集 | 自发运动减少 | NA |
| A30P+A53T[ | Thy-1 | C57BL/6J小鼠 | 约50% | 包涵体 | 自发运动减少 | NA |
| A53T[ | PrP | C57BL/6J小鼠 | 约40% | 异常聚集 | 自发运动和在棒时间减少、步幅缩短 | NA |
| Wild type(SNCA-OVX line)[ | PrP | C57BL/6J×B6小鼠 | 约30% | 异常聚集 | 在棒时间减少,前爪步幅缩短 | 粪便重量增加 |
| Wild type[ | PrP | SD大鼠 | 约40% | 包涵体 | 自发活动和直立活动减少 | 嗅觉障碍 |
Table 2 Characteristics of transgenic mouse PD models with different α-synuclein
α突触核蛋白基因 α-synuclein | 启动子 Promoter | 品种品系 Breed and strain | 多巴胺能神经元的损失率 Loss rate of dopaminergic neurons | α突触核蛋白的病理表型 Pathological phenotype of α-synuclein | 运动表型 Motor phenotype | 非运动表型 Non-motor phenotype |
|---|---|---|---|---|---|---|
| Gene fragments(1~130)[ | Thy-1 | C57BL/6J小鼠 | 约50% | 异常聚集 | 自发运动减少 | NA |
| A30P+A53T[ | Thy-1 | C57BL/6J小鼠 | 约50% | 包涵体 | 自发运动减少 | NA |
| A53T[ | PrP | C57BL/6J小鼠 | 约40% | 异常聚集 | 自发运动和在棒时间减少、步幅缩短 | NA |
| Wild type(SNCA-OVX line)[ | PrP | C57BL/6J×B6小鼠 | 约30% | 异常聚集 | 在棒时间减少,前爪步幅缩短 | 粪便重量增加 |
| Wild type[ | PrP | SD大鼠 | 约40% | 包涵体 | 自发活动和直立活动减少 | 嗅觉障碍 |
模型类型 Model type | 建模成功时间 Time to successful model establishment | 损伤部位 Site of injury | 路易体 Lewy body | 成模检测时间 Detection time | 运动行为测试方法 Motor behavior testing method | 可重复性 Repeatability | 应用范围 Scope of application |
|---|---|---|---|---|---|---|---|
6-OHDA大鼠PD模型 6-OHDA rat PD model | 给药后1~3周内模型稳定 | 黑质致密部和纹状体 | 无 | 2~4周 | 旷场,转圈,转棒,步态运动明显减少 | 高(旋转行为与神经元损伤程度高度一致,成功率>90%) | DBS、药物、干细胞治疗干预 |
MPTP小鼠PD模型 MPTP mouse PD model | 给药后1~2周内模型稳定 | 黑质致密部和纹状体 | 无 | 2~4周 | 旷场,转棒,爬杆,步态运动明显减少 | 中(行为学测试结果可能存在个体差异) | DBS、药物、干细胞治疗干预 |
SNCA转基因大鼠PD模型 SNCA transgenic rat PD model | 3个月以上 | 根据转基因类型而定,可影响多个脑区 | 有 | 出生后数周至数月 | 旷场,转棒,步态运动减少 | 高(转基因技术可重复性强) | 基因治疗、药物干预 |
Parkin基因敲除小鼠PD模型 Parkin gene knockout mouse PD model | 3个月以上 | 根据转基因类型而定,可能影响多个脑区 | 无 | 出生后数周 | 旷场,爬杆,步态运动减少 | 高(转基因技术可重复性强) | 基因治疗、药物干预 |
Table 3 Comparison of characteristics of several commonly used rat and mouse models of Parkinson disease
模型类型 Model type | 建模成功时间 Time to successful model establishment | 损伤部位 Site of injury | 路易体 Lewy body | 成模检测时间 Detection time | 运动行为测试方法 Motor behavior testing method | 可重复性 Repeatability | 应用范围 Scope of application |
|---|---|---|---|---|---|---|---|
6-OHDA大鼠PD模型 6-OHDA rat PD model | 给药后1~3周内模型稳定 | 黑质致密部和纹状体 | 无 | 2~4周 | 旷场,转圈,转棒,步态运动明显减少 | 高(旋转行为与神经元损伤程度高度一致,成功率>90%) | DBS、药物、干细胞治疗干预 |
MPTP小鼠PD模型 MPTP mouse PD model | 给药后1~2周内模型稳定 | 黑质致密部和纹状体 | 无 | 2~4周 | 旷场,转棒,爬杆,步态运动明显减少 | 中(行为学测试结果可能存在个体差异) | DBS、药物、干细胞治疗干预 |
SNCA转基因大鼠PD模型 SNCA transgenic rat PD model | 3个月以上 | 根据转基因类型而定,可影响多个脑区 | 有 | 出生后数周至数月 | 旷场,转棒,步态运动减少 | 高(转基因技术可重复性强) | 基因治疗、药物干预 |
Parkin基因敲除小鼠PD模型 Parkin gene knockout mouse PD model | 3个月以上 | 根据转基因类型而定,可能影响多个脑区 | 无 | 出生后数周 | 旷场,爬杆,步态运动减少 | 高(转基因技术可重复性强) | 基因治疗、药物干预 |
特性 Characteristic | 猕猴 Macaque | 食蟹猴 Cynomolgus monkey | 狨猴 Marmoset |
|---|---|---|---|
MPTP敏感性(ED50值) MPTP sensitivity (ED50 value) | 猕猴对MPTP的敏感性存在个体差异。常用肌内注射MPTP的剂量从0.2 mg/kg开始,逐渐增加至0.5 mg/kg | 食蟹猴对MPTP的敏感性存在个体差异,且中老年食蟹猴对MPTP的敏感性更高。颈内动脉注射2~3 mg MPTP后辅以静脉注射(≤0.4 mg/kg) | 狨猴对MPTP的敏感性相对较低,通常需要更高的剂量(如每周0.5 mg/kg,连续5周)才能诱导出PD症状 |
行为学表型差异 Behavioral phenotype differences | 会出现静止性震颤、姿势/意向性震颤、运动迟缓、步态不稳、面部表情减少、姿势僵硬等典型症状,还可能伴有嗜睡、打哈欠、便秘、生活自理能力下降、发声减少等非典型症状 | 会出现明显的PD症状,如运动迟缓、震颤等,且症状的严重程度和进展速度因个体而异 | 会出现运动迟缓、震颤等症状,但症状的严重程度相对较低 |
成本评估 Cost assessment | 猕猴体型较大,饲养成本较高,且该动物对MPTP的敏感性存在个体差异,建模过程可能需要更多的时间和精力来调整给药方案 | 食蟹猴体型相对较小,饲养成本略低于猕猴,但其对MPTP的敏感性存在个体差异,建模过程中需要根据个体情况调整给药方案 | 狨猴体型最小,饲养成本相对较低,但需要更高的剂量才能诱导出症状,可能会增加药物成本 |
Table 4 Main characteristics of Parkinson disease models in macaques, cynomolgus monkeys, and marmosets
特性 Characteristic | 猕猴 Macaque | 食蟹猴 Cynomolgus monkey | 狨猴 Marmoset |
|---|---|---|---|
MPTP敏感性(ED50值) MPTP sensitivity (ED50 value) | 猕猴对MPTP的敏感性存在个体差异。常用肌内注射MPTP的剂量从0.2 mg/kg开始,逐渐增加至0.5 mg/kg | 食蟹猴对MPTP的敏感性存在个体差异,且中老年食蟹猴对MPTP的敏感性更高。颈内动脉注射2~3 mg MPTP后辅以静脉注射(≤0.4 mg/kg) | 狨猴对MPTP的敏感性相对较低,通常需要更高的剂量(如每周0.5 mg/kg,连续5周)才能诱导出PD症状 |
行为学表型差异 Behavioral phenotype differences | 会出现静止性震颤、姿势/意向性震颤、运动迟缓、步态不稳、面部表情减少、姿势僵硬等典型症状,还可能伴有嗜睡、打哈欠、便秘、生活自理能力下降、发声减少等非典型症状 | 会出现明显的PD症状,如运动迟缓、震颤等,且症状的严重程度和进展速度因个体而异 | 会出现运动迟缓、震颤等症状,但症状的严重程度相对较低 |
成本评估 Cost assessment | 猕猴体型较大,饲养成本较高,且该动物对MPTP的敏感性存在个体差异,建模过程可能需要更多的时间和精力来调整给药方案 | 食蟹猴体型相对较小,饲养成本略低于猕猴,但其对MPTP的敏感性存在个体差异,建模过程中需要根据个体情况调整给药方案 | 狨猴体型最小,饲养成本相对较低,但需要更高的剂量才能诱导出症状,可能会增加药物成本 |
| [1] | COSTA H N, ESTEVES A R, EMPADINHAS N, et al. Parkinson's disease: a multisystem disorder[J]. Neurosci Bull, 2023, 39(1):113-124. DOI:10.1007/s12264-022-00934-6 . |
| [2] | LEES A J, HARDY J, REVESZ T. Parkinson's disease[J]. Lancet, 2009, 373(9680):2055-2066. DOI:10.1016/S0140-6736(09)60492-X . |
| [3] | YARIBASH S, MOHAMMADI K, SANI M A. Alpha-synuclein pathophysiology in neurodegenerative disorders: a review focusing on molecular mechanisms and treatment advances in Parkinson's disease[J]. Cell Mol Neurobiol, 2025, 45(1): 30. DOI: 10.1007/s10571-025-01544-2 . |
| [4] | MORRIS H R, SPILLANTINI M G, SUE C M, et al. The pathogenesis of Parkinson's disease[J]. Lancet, 2024, 403(10423): 293-304. DOI: 10.1016/S0140-6736(23)01478-2 . |
| [5] | BARKER R A, DROUIN-OUELLET J, PARMAR M. Cell-based therapies for Parkinson disease: past insights and future potential[J]. Nat Rev Neurol, 2015, 11(9): 492-503. DOI: 10.1038/nrneurol.2015.123 . |
| [6] | FREED C R, GREENE P E, BREEZE R E, et al. Transplantation of embryonic dopamine neurons for severe Parkinson's disease[J]. N Engl J Med, 2001, 344(10):710-719. DOI: 10.1056/NEJM200103083441002 . |
| [7] | KRIKS S, SHIM J W, PIAO J H, et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson's disease[J]. Nature, 2011, 480(7378): 547-551. DOI: 10.1038/nature10648 . |
| [8] | HAN F B, HU B Y. Stem cell therapy for Parkinson's disease[J]. Adv Exp Med Biol, 2020, 1266: 21-38. DOI: 10.1007/978-981-15-4370-8_3 . |
| [9] | SCHWEITZER J S, SONG B, HERRINGTON T M, et al. Personalized iPSC-derived dopamine progenitor cells for Parkinson's disease[J]. N Engl J Med, 2020, 382(20): 1926-1932. DOI: 10.1056/NEJMoa1915872 . |
| [10] | MORIZANE A. Cell therapy for Parkinson's disease with induced pluripotent stem cells[J]. Inflamm Regen, 2023, 43(1): 16. DOI: 10.1186/s41232-023-00269-3 . |
| [11] | KALIA L V, LANG A E. Parkinson's disease[J]. Lancet, 2015, 386(9996):896-912. DOI: 10.1016/S0140-6736(14)61393-3 . |
| [12] | TANNER C M, KAMEL F, ROSS G W, et al. Rotenone, paraquat, and Parkinson's disease[J]. Environ Health Perspect, 2011, 119(6):866-872. DOI: 10.1289/ehp.1002839 . |
| [13] | HAN F B, HU B Y. Stem cell therapy for Parkinson's disease[J]. Adv Exp Med Biol, 2020, 1266:21-38. DOI: 10.1007/978-981-15-4370-8_3 . |
| [14] | YE H, ROBAK L A, YU M G, et al. Genetics and pathogenesis of Parkinson's syndrome[J]. Annu Rev Pathol, 2023, 18:95-121. DOI: 10.1146/annurev-pathmechdis-031521-034145 . |
| [15] | JIA F Z, FELLNER A, KUMAR K R. Monogenic Parkinson's disease: genotype, phenotype, pathophysiology, and genetic testing[J]. Genes, 2022, 13(3):471. DOI: 10.3390/genes13030471 . |
| [16] | BLOEM B R, OKUN M S, KLEIN C. Parkinson's disease[J]. Lancet, 2021, 397(10291):2284-2303. DOI: 10.1016/S0140-6736(21)00218-X . |
| [17] | HAN F B, GRIMES D A, LI F, et al. Mutations in the glucocerebrosidase gene are common in patients with Parkinson's disease from Eastern Canada[J]. Int J Neurosci, 2016, 126(5):415-421. DOI: 10.3109/00207454.2015.1023436 . |
| [18] | SONG N, WANG W, CHEN C, et al. Analysis of SMPD1 gene mutations in Chinese patients with Parkinson's disease[J]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi, 2018, 35(3):319-323. DOI: 10.3760/cma.j.issn.1003-9406.2018.03.003 . |
| [19] | BAEK M, CHOE Y J, BANNWARTH S, et al. TDP-43 and PINK1 mediate CHCHD10S59L mutation-induced defects in Drosophila and in vitro [J]. Nat Commun, 2021, 12(1):1924. DOI: 10.1038/s41467-021-22145-9 . |
| [20] | SONG N, WANG Y, ZHOU L X, et al. Genetic analysis of the LRP10 gene in Chinese patients with Parkinson's disease[J]. Neurol Sci, 2023, 44(3):905-912. DOI: 10.1007/s10072-022-06496-9 . |
| [21] | LIN C H, TSAI P I, LIN H Y, et al. Mitochondrial UQCRC1 mutations cause autosomal dominant Parkinsonism with polyneuropathy[J]. Brain, 2020, 143(11):3352-3373. DOI: 10. 1093/brain/awaa279 . |
| [22] | DENG H, FAN K, JANKOVIC J. The role of TMEM230 gene in Parkinson's disease[J]. J Parkinsons Dis, 2018, 8(4):469-477. DOI: 10.3233/JPD-181421 . |
| [23] | WANG X B, WHELAN E, LIU Z H, et al. Controversy of TMEM230 associated with Parkinson's disease[J]. Neuroscience, 2021, 453:280-286. DOI: 10.1016/j.neuroscience. 2020.11.004 . |
| [24] | LANG A E, LOZANO A M. Parkinson's disease[J]. N Engl J Med, 1998, 339(16):1130-1143. DOI: 10.1056/nejm19981015339 1607 . |
| [25] | GERFEN C R. Indirect-pathway neurons lose their spines in Parkinson disease[J]. Nat Neurosci, 2006, 9(2):157-158. DOI: 10.1038/nn0206-157 . |
| [26] | MCGREGOR M M, NELSON A B. Circuit mechanisms of Parkinson's disease[J]. Neuron, 2019, 101(6):1042-1056. DOI: 10.1016/j.neuron.2019.03.004 . |
| [27] | CHU H Y. Synaptic and cellular plasticity in Parkinson's disease[J]. Acta Pharmacol Sin, 2020, 41(4):447-452. DOI: 10.1038/s41401-020-0371-0 . |
| [28] | CERNERA S, EISINGER R S, WONG J K, et al. Long-term Parkinson's disease quality of life after staged DBS: STN vs GPi and first vs second lead[J]. NPJ Parkinsons Dis, 2020, 6:13. DOI: 10.1038/s41531-020-0115-3 . |
| [29] | TOLOSA E, GARRIDO A, SCHOLZ S W, et al. Challenges in the diagnosis of Parkinson's disease[J]. Lancet Neurol, 2021, 20(5):385-397. DOI: 10.1016/S1474-4422(21)00030-2 . |
| [30] | NABIZADEH F, PIRAHESH K, KHALILI E. Olfactory dysfunction is associated with motor function only in tremor-dominant Parkinson's disease[J]. Neurol Sci, 2022, 43(7):4193-4201. DOI: 10.1007/s10072-022-05952-w . |
| [31] | UEMURA N, UEMURA M T, LUK K C, et al. Cell-to-cell transmission of tau and α-synuclein[J]. Trends Mol Med, 2020, 26(10):936-952. DOI: 10.1016/j.molmed.2020.03.012 . |
| [32] | ELDEEB M A, THOMAS R A, RAGHEB M A, et al. Mitochondrial quality control in health and in Parkinson's disease[J]. Physiol Rev, 2022, 102(4):1721-1755. DOI: 10.1152/physrev.00041.2021 . |
| [33] | HU S L, TAN J Q, QIN L X, et al. Molecular chaperones and Parkinson's disease[J]. Neurobiol Dis, 2021, 160:105527. DOI: 10.1016/j.nbd.2021.105527 . |
| [34] | WANG F J, ZHANG G J, ZHAI Q F. Role and mechanism of molecular hydrogen in the treatment of Parkinson's diseases[J]. Front Neurosci, 2025, 19:1576773. DOI: 10.3389/fnins. 2025.1576773 . |
| [35] | NECHUSHTAI L, FRENKEL D, PINKAS-KRAMARSKI R. Autophagy in Parkinson's disease[J]. Biomolecules, 2023, 13(10):1435. DOI: 10.3390/biom13101435 . |
| [36] | ARAÚJO B, CARIDADE-SILVA R, SOARES-GUEDES C, et al. Neuroinflammation and Parkinson's disease—from neurode-generation to therapeutic opportunities[J]. Cells, 2022, 11(18):2908. DOI: 10.3390/cells11182908 . |
| [37] | CHIA S J, TAN E K, CHAO Y X. Historical perspective: models of Parkinson's disease[J]. Int J Mol Sci, 2020, 21(7):2464. DOI: 10.3390/ijms21072464 . |
| [38] | MUSTAPHA M, TAIB C N MAT. MPTP-induced mouse model of Parkinson's disease: a promising direction of therapeutic strategies[J]. Bosn J Basic Med Sci, 2021, 21(4):422-433. DOI: 10.17305/bjbms.2020.5181 . |
| [39] | 胡毅龙, 赵怡楠, 苗晋鑫, 等. 大小鼠帕金森病行为学评价方法概述及常用动物模型特点分析[J]. 中国实验动物学报, 2024, 32(7): 942-954. DOI: 10.3969/j.issn.1005-4847.2024.07.013 . |
| HU Y L, ZHAO Y N, MIAO J X, et al. Overview of behavioral evaluation methods of Parkinson's disease in rat and mouse and analysis of the characteristics of common animal models[J]. Acta Lab Anim Sci Sin, 2024, 32(7): 942-954. DOI: 10.3969/j.issn.1005-4847.2024.07.013 . | |
| [40] | KWON H S, KOH S H. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes[J]. Transl Neurodegener, 2020, 9(1):42. DOI: 10.1186/s40035-020-00221-2 . |
| [41] | ZHANG Q S, HENG Y, MOU Z, et al. Reassessment of subacute MPTP-treated mice as animal model of Parkinson's disease[J]. Acta Pharmacol Sin, 2017, 38(10):1317-1328. DOI: 10.1038/aps.2017.49 . |
| [1] | LI Yuan, ZHANG Mei-ying. Research Progress on Autophagy with Parkinson Disease and Related Models [J]. Laboratory Animal and Comparative Medicine, 2015, 35(4): 335-340. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||