
Laboratory Animal and Comparative Medicine ›› 2026, Vol. 46 ›› Issue (1): 127-137.DOI: 10.12300/j.issn.1674-5817.2025.072
• Laboratory Animal Welfare and Ethics • Previous Articles Next Articles
LI Xiaoqin1, YU Wenlan2, DUAN Yizhu1, LIU Zhonghua2, WU Guodong1, SHI Wenqi1, FU Hongkun3( )(
)( )
)
Received:2025-05-15
Revised:2025-08-20
Online:2026-02-25
Published:2026-02-14
Contact:
FU Hongkun
CLC Number:
LI Xiaoqin,YU Wenlan,DUAN Yizhu,et al. Construction of Laboratory Animal Science and Technology Ethics Governance System in China and Its Preliminary Practice in Guangdong Province[J]. Laboratory Animal and Comparative Medicine, 2026, 46(1): 127-137. DOI: 10.12300/j.issn.1674-5817.2025.072.
Add to citation manager EndNote|Ris|BibTeX
URL: https://www.slarc.org.cn/dwyx/EN/10.12300/j.issn.1674-5817.2025.072
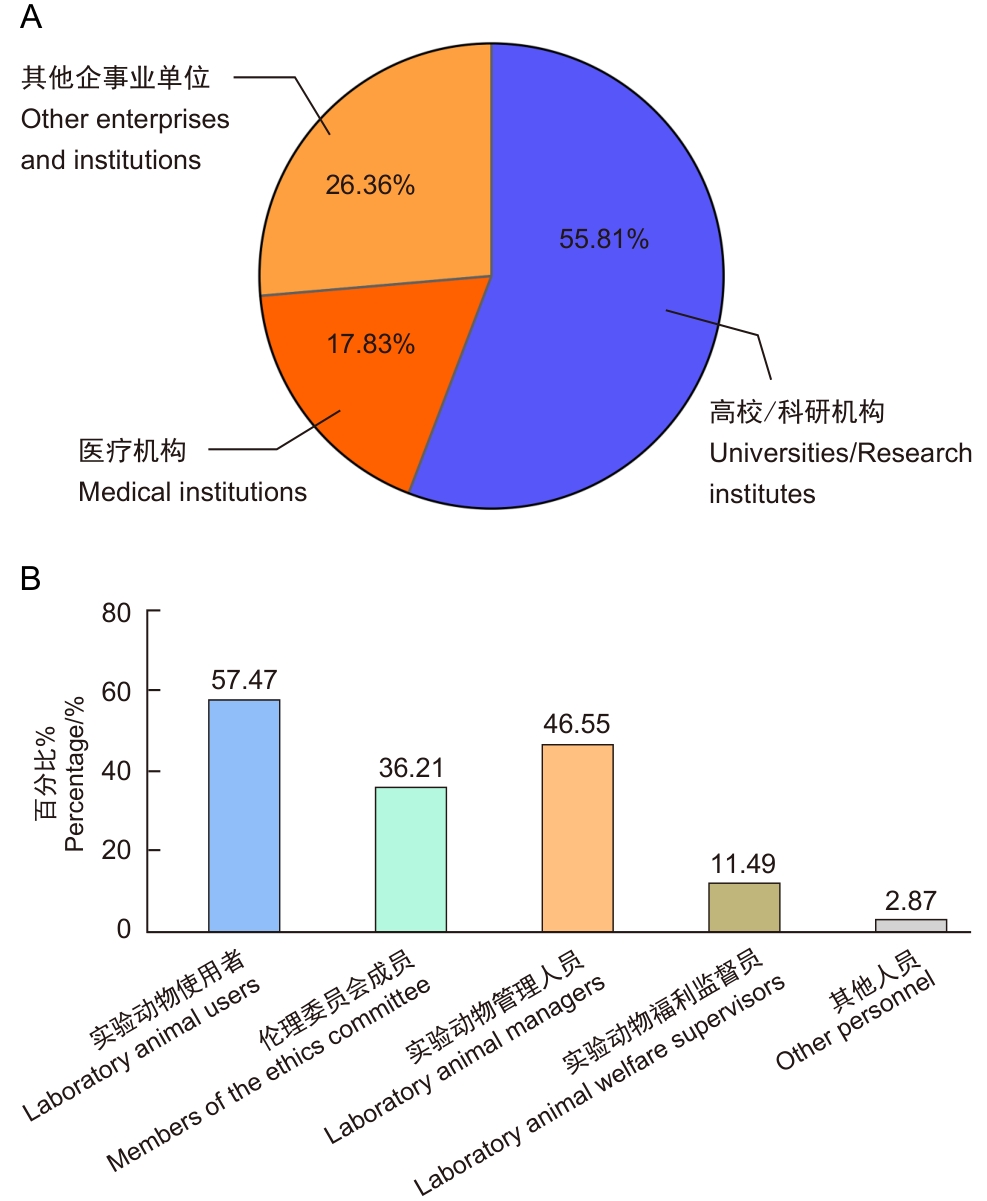
Figure 1 Proportional distribution of survey subject types
调查事项 Investigation items | 主要调查内容 Main survey contents | 选择“是”占比/% Percentage of "yes" responses/% | |||
|---|---|---|---|---|---|
总体 Overall | 高校/科研机构 Universities/research institutes | 医疗机构 Medical institutions | 其他企事业单位 Other enterprises and institutions | ||
制度保障 Institutional guarantee | 是否建立伦理委员会工作章程或相关制度 | 98.28 | 97.94 | 100.00 | 97.87 |
| 是否建立审查标准操作流程 | 81.61 | 75.26 | 86.67 | 91.49 | |
| 是否建立项目跟踪监督机制 | 66.09 | 67.01 | 63.33 | 65.96 | |
| 是否建立投诉处理渠道 | 52.87 | 58.76 | 40.00 | 48.94 | |
| 是否建立实验动物福利伦理(审查)档案 | 96.55 | 97.94 | 100.00 | 91.49 | |
责任体系 Responsibility system | 是否设立实验动物福利伦理审查机构 | 88.51 | 85.57 | 86.67 | 97.87 |
| 伦理委员会成员数量及构成是否符合指南要求 | 89.66 | 87.63 | 96.67 | 89.36 | |
| 伦理委员会任期是否固定 | 47.70 | 41.24 | 50.00 | 59.57 | |
审查监管 Review and supervision | 实验动物使用是否严格执行“3R”原则 | 91.95 | 92.78 | 93.33 | 89.36 |
| 是否对违法违规现象进行调查 | 89.66 | 88.66 | 90.00 | 91.49 | |
| 是否在项目结束时进行终结审查 | 78.74 | 81.44 | 73.33 | 76.60 | |
| 伦理审查是否严格遵守福利原则 | 94.25 | 93.81 | 90.00 | 97.87 | |
| 伦理审查申请书填写是否困难 | 13.79 | 14.43 | 13.33 | 12.77 | |
| 是否认为存在监管机制不健全问题 | 47.13 | 44.33 | 53.33 | 48.94 | |
| 是否认为存在审查流于形式问题 | 47.13 | 47.42 | 36.67 | 53.19 | |
教育培训 Education and training | 是否建立定期培训制度 | 79.89 | 80.41 | 86.67 | 74.47 |
| 是否认为存在伦理意识淡薄问题 | 55.17 | 55.67 | 46.67 | 59.57 | |
| 是否认为缺乏实验动物科技伦理专业培训 | 60.34 | 57.73 | 60.00 | 68.08 | |
Table 1 Questionnaire survey on laboratory animal science and technology ethics management of laboratory animal production or use units in Guangdong Province
调查事项 Investigation items | 主要调查内容 Main survey contents | 选择“是”占比/% Percentage of "yes" responses/% | |||
|---|---|---|---|---|---|
总体 Overall | 高校/科研机构 Universities/research institutes | 医疗机构 Medical institutions | 其他企事业单位 Other enterprises and institutions | ||
制度保障 Institutional guarantee | 是否建立伦理委员会工作章程或相关制度 | 98.28 | 97.94 | 100.00 | 97.87 |
| 是否建立审查标准操作流程 | 81.61 | 75.26 | 86.67 | 91.49 | |
| 是否建立项目跟踪监督机制 | 66.09 | 67.01 | 63.33 | 65.96 | |
| 是否建立投诉处理渠道 | 52.87 | 58.76 | 40.00 | 48.94 | |
| 是否建立实验动物福利伦理(审查)档案 | 96.55 | 97.94 | 100.00 | 91.49 | |
责任体系 Responsibility system | 是否设立实验动物福利伦理审查机构 | 88.51 | 85.57 | 86.67 | 97.87 |
| 伦理委员会成员数量及构成是否符合指南要求 | 89.66 | 87.63 | 96.67 | 89.36 | |
| 伦理委员会任期是否固定 | 47.70 | 41.24 | 50.00 | 59.57 | |
审查监管 Review and supervision | 实验动物使用是否严格执行“3R”原则 | 91.95 | 92.78 | 93.33 | 89.36 |
| 是否对违法违规现象进行调查 | 89.66 | 88.66 | 90.00 | 91.49 | |
| 是否在项目结束时进行终结审查 | 78.74 | 81.44 | 73.33 | 76.60 | |
| 伦理审查是否严格遵守福利原则 | 94.25 | 93.81 | 90.00 | 97.87 | |
| 伦理审查申请书填写是否困难 | 13.79 | 14.43 | 13.33 | 12.77 | |
| 是否认为存在监管机制不健全问题 | 47.13 | 44.33 | 53.33 | 48.94 | |
| 是否认为存在审查流于形式问题 | 47.13 | 47.42 | 36.67 | 53.19 | |
教育培训 Education and training | 是否建立定期培训制度 | 79.89 | 80.41 | 86.67 | 74.47 |
| 是否认为存在伦理意识淡薄问题 | 55.17 | 55.67 | 46.67 | 59.57 | |
| 是否认为缺乏实验动物科技伦理专业培训 | 60.34 | 57.73 | 60.00 | 68.08 | |
| [1] | 中共中央办公厅, 国务院办公厅. 关于加强科技伦理治理的意见[A/OL]. (2022-03-20)[2025-04-30]. . |
| General Office of the CPC Central Committee, General Office of the State Council. Opinions on enhancing governance over ethics in science and technology[A/OL]. (2022-03-20)[2025-04-30]. . | |
| [2] | 科学技术部.关于印发«科技伦理审查办法(试行)»的通知: 国科发监〔2023〕167号[A/OL]. (2023-10-08)[2025-04-30]. . |
| Ministry of Science and Technology. Notice on Issuing the "Measures for the Review of Science and Technology Ethics (Trial)": Guo Ke Fa Jian [2023] No. 167 [A/OL]. (2023-10-08)[2025-04-30]. . | |
| [3] | AHN N, PARK J, ROH S. Use of laboratory animals and issues regarding the procurement of animals for research in Korea[J]. Lab Anim Res, 2023, 39(1):10. DOI:10.1186/s42826-023-00161-8 . |
| [4] | RESHETNYKOV M, STUMM K, ZWICK M, et al. Decision trees for determining the fate of laboratory animals[J]. Lab Anim, 2025, 59(2):157-168. DOI:10.1177/00236772241271034 . |
| [5] | TANNENBAUM J, BENNETT B T. Russell and Burch's 3Rs then and now: the need for clarity in definition and purpose[J]. J Am Assoc Lab Anim Sci, 2015, 54(2):120-132. |
| [6] | FRANCO N H, KERTON A, LEWIS D I. Education in laboratory animal science and the 3Rs[J]. Lab Anim, 2023, 57(2):109-111. DOI:10.1177/00236772231162166 . |
| [7] | GRIMM H, BILLER-ANDORNO N, BUCH T, et al. Advancing the 3Rs: innovation, implementation, ethics and society[J]. Front Vet Sci, 2023, 10:1185706. DOI:10.3389/fvets.2023.1185706 . |
| [8] | 黄术兵. 中西实验动物伦理和福利发展比较研究[J]. 医学与哲学, 2020, 41(17):33-38. DOI:10.12014/j.issn.1002-0772.2020.17.09 . |
| HUANG S B. Comparative study on ethics and welfare of laboratory animals between China and the west countries[J]. Med Philos, 2020, 41(17):33-38. DOI:10.12014/j.issn.1002-0772.2020.17.09 . | |
| [9] | 吴孝槐, 肖巧喆, 庞万勇, 等. CNAS实验动物机构认可与AAALAC认证的比较分析[J]. 实验动物与比较医学, 2022, 42(3):237-243. DOI: 10.12300/j.issn.1674-5817.2022.033 . |
| WU X H, XIAO Q Z, PANG W Y, et al. A comparative analysis of CNAS laboratory animal institutions accreditation and AAALAC certification[J]. Lab Anim Comp Med, 2022, 42(3): 237-243. DOI: 10.12300/j.issn.1674-5817.2022.033 . | |
| [10] | 刘朝霞, 林惠然. 关于完善科研机构实验动物福利伦理管理的对策建议[J]. 中国动物检疫, 2024, 41(2): 31-35. DOI:10.3969/j.issn.1005-944X.2024.02.007 . |
| LIU Z X, LIN H R. Countermeasures on improving the management of experimental animal welfare and ethics in scientific institutions[J]. China Anim Health Insp, 2024, 41(2): 31-35. DOI:10.3969/j.issn.1005-944X.2024.02.007 . | |
| [11] | 郇长超, 胡娇, 高崧. 实验动物福利相关问题及改善措施[J]. 动物医学进展, 2024, 45(12):113-116. DOI:10.16437/j.cnki.1007-5038.2024.12.002 . |
| HUAN C C, HU J, GAO S. Laboratory animal welfare related issues and improvement measures[J]. Prog Vet Med, 2024, 45(12):113-116. DOI:10.16437/j.cnki.1007-5038.2024.12.002 . | |
| [12] | 李夏莹, 田永路, 韦玉生, 等. 实验动物福利伦理审查要点[J]. 中国实验动物学报, 2025, 33(4): 616-622. DOI:10.3969/j.issn.1005-4847.2025.04.015 . |
| LI X Y, TIAN Y L, WEI Y S, et al. Key aspects for the ethical review of laboratory animal welfare protocols[J]. Acta Lab Anim Sci Sin, 2025, 33(4): 616-622. DOI:10.3969/j.issn.1005-4847.2025.04.015 . | |
| [13] | 赵勇. 动物实验伦理的三个维度: 基于生命价值、动物福利和风险防范的阐析[J]. 实验动物与比较医学, 2024, 44(4): 445-454. DOI:10.12300/j.issn.1674-5817.2024.108 . |
| ZHAO Y. Three dimensions of animal experiment ethics: analysis based on value of life, animal welfare, and risk prevention[J]. Lab Anim Comp Med, 2024, 44(4): 445-454. DOI:10.12300/j.issn.1674-5817.2024.108 . | |
| [14] | 闵凡贵, 富宏坤, 刘永刚, 等.感染性动物实验的福利与伦理特殊要求[J].实验动物与比较医学, 2025, 45(2): 239-246. DOI: 10.12300/j.issn.1674-5817.2024.122 . |
| MIN F G, FU H K, LIU Y G, et al. Special welfare and ethical requirements for infectious animal experiments[J]. Lab Anim Comp Med, 2025, 45(2): 239-246. DOI: 10.12300/j. issn. 1674-5817. 2024.122 . | |
| [15] | 李彪, 余亮, 陈梅丽, 等. 广东省实验动物许可证管理工作的现状及对策[J]. 中国比较医学杂志, 2014, 24(6): 73-77. DOI:10.3969/j.issn.1671.7856.2014.006.016 . |
| LI B, YU L, CHEN M L, et al. Present situation and strategy of laboratory animal license management in Guangdong Province[J]. Chin J Comp Med, 2014, 24(6): 73-77. DOI:10.3969/j.issn.1671.7856.2014.006.016 . | |
| [16] | 李会萍, 梁楚军, 赵维波. 广东省实验动物许可证管理现状[J]. 实验动物与比较医学, 2022, 42(6): 577-582. DOI:10.12300/j.issn.1674-5817.2022.107 . |
| LI H P, LIANG C J, ZHAO W B. The current status of laboratory animal license administration in Guangdong Province[J]. Lab Anim Comp Med, 2022, 42(6): 577-582. DOI:10.12300/j.issn.1674-5817.2022.107 . | |
| [17] | 邓少嫦, 林丹荣, 梁楚军, 等. «广东省实验动物管理条例»制度特点及近十年实施情况分析[J]. 实验动物与比较医学, 2024, 44(4): 455-462. DOI:10.12300/j.issn.1674-5817.2024.030 . |
| DENG S C, LIN D R, LIANG C J, et al. Analysis of institutional characteristics and implementation of Guangdong Province laboratory animals ordinance in the past decade[J]. Lab Anim Comp Med, 2024, 44(4): 455-462. DOI:10.12300/j.issn.1674-5817.2024.030 . | |
| [18] | 国家科学技术委员会. 实验动物管理条例(第3次修订)[A/OL]. (2017-03-01)[2025-04-30]. . |
| National Science and Technology Commission. Regulations on the administration of laboratory animals (Third Amendment)[A/OL]. (2017-03-01)[2025-04-30]. . | |
| [19] | 科学技术部. 关于善待实验动物的指导性意见: 国科发财字〔2006〕398号[A/OL]. (2006-09-30)[2025-04-30]. . |
| Ministry of Science and Technology. Guiding opinions on treating experimental animals well: Guo Ke Fa Cai Zi [2006] No. 398 [A/OL]. (2006-09-30)[2025-04-30]. . | |
| [20] | 国家质量监督检验检疫总局, 国家标准化管理委员会. 实验动物 福利伦理审查指南: [S]. 北京: 中国标准出版社, 2018. |
| General Administration of Quality Supervision, Inspection and Quarantine, National Standardization Administration. Laboratory animal—Guideline for ethical review of animal welfare: [S]. Beijing: Standards Press of China, 2018. | |
| [21] | 国家市场监督管理总局, 国家标准化管理委员会. 实验动物 福利通则: [S]. 北京: 中国标准出版社, 2022. |
| State Administration for Market Regulation, National Standardization Administration. Laboratory animals—General code of animal welfare: [S]. Beijing: Standards Press of China, 2022. | |
| [22] | 国家认证认可监督管理委员会. 实验动物安乐死技术规范: RB/T 061—2021 [S]. 北京:中国标准出版社, 2021. |
| National Certification and Accreditation Administration. Technical specifications on euthanasia of laboratory animals: RB/T 061—2021 [S]. Beijing: Standards Press of China, 2021. | |
| [23] | 中国实验动物学会. 实验动物 福利伦理委员会工作指南: T/CALAS 73—2019 [S]. 北京: 中国标准出版社, 2019. |
| Chinese Association for Laboratory Animal Sciences. Laboratory animals—Work specification of the welfare and ethics committee: T/CALAS 73—2019 [S]. Beijing: Standards Press of China, 2019. | |
| [24] | 农业农村部. 动物诊疗机构管理办法[A/OL]. (2022-09-07)[2025-04-30]. . |
| Ministry of Agriculture and Rural Affairs. Management Method for Animal Clinics[A/OL]. (2022-09-07)[2025-04-30]. . | |
| [25] | 农业农村部. 动物检疫管理办法[A/OL]. (2022-09-07)[2025-04-30]. . |
| Ministry of Adriculture and Rural Affairs. Management Method for Animal quarantine[A/OL]. (2022-09-07)[2025-04-30]. . | |
| [26] | 国家质量监督检验检疫总局, 国家标准化管理委员会. 实验动物机构 质量和能力的通用要求: [S]. 北京: 中国标准出版社, 2014. |
| General Administration of Quality Supervision, Inspection and Quarantine, National Standardization Administration. Laboratory animal institutions—General requirements for quality and competence: [S]. Beijing: Standards Press of China, 2014. | |
| [27] | ZHANG W, XIE Z, FANG X, et al. Laboratory animal ethics education improves medical students' awareness of laboratory animal ethics[J]. BMC Med Educ, 2024, 24(1):709. DOI:10.1186/s12909-024-05703-9 . |
| [28] | 广东省人民政府. 广东省实验动物管理条例[A/OL]. (2022-02-07)[2025-04-30]. . |
| People's Government of Guangdong Province. Regulations of Guangdong Province on the administration of laboratory animals[A/OL]. (2022-02-07)[2025-04-30]. . | |
| [29] | 宋应登, 霍竹, 邓益志. 英国科技伦理监管体系研究与启示[J]. 中国科技论坛, 2024(8):179-188. DOI:10.13580/j.cnki.fstc.2024.08.012 . |
| SONG Y D, HUO Z, DENG Y Z. Research on UK governance system of science and technology ethics and implication[J]. Forum Sci Technol China, 2024(8):179-188. DOI:10.13580/j.cnki.fstc.2024.08.012 . | |
| [30] | 北京市市场监督管理局. 实验动物福利伦理审查技术规范: [S/OL]. (2020-06-30)[2025-04-30]. . |
| Beijing Municipal Market Supervision Administration. Technical specification of ethical review for laboratory animal welfare: [S/OL]. (2020-06-30)[2025-04-30]. . |
| [1] | DENG Shaochang, LIN Danrong, LIANG Chujun, LEI Weiqiao, YANG Jinchun, ZHAO Weibo. Analysis of Institutional Characteristics and Implementation of Guangdong Province Laboratory Animals Ordinance in the Past Decade [J]. Laboratory Animal and Comparative Medicine, 2024, 44(4): 455-462. |
| [2] | Huiping LI, Chujun LIANG, Weibo ZHAO. The Current Status of Laboratory Animal License Administration in Guangdong Province [J]. Laboratory Animal and Comparative Medicine, 2022, 42(6): 577-582. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||